 | ||
The Frasch process is a method to extract sulfur from underground deposits. It is the only economic method of recovering sulfur from elemental deposits. Most of the world's sulfur was obtained this way until the late 20th century, when sulfur recovered from petroleum and gas sources (recovered sulfur) became more commonplace (see Claus process).
Contents
In the Frasch process, superheated water is pumped into the sulfur deposit; the sulfur melts and is extracted. The Frasch process is able to produce high purity sulfur.
History
In 1867, miners discovered sulfur in the caprock of a salt dome in Calcasieu Parish, Louisiana, but it was beneath quicksand, which prevented mining. In 1894 the German-born American chemist, Herman Frasch (1852–1914), devised his Frasch method of sulfur removal using pipes to bypass the quicksand. The process proved successful, on December 24, 1894, when the first molten sulfur was brought to the surface. However, the high cost of fuel needed to heat the water made the process uneconomic until the 1901 discovery of the Spindletop oil field in Texas provided cheap fuel oil to the region. The Frasch process began economic production at Sulphur Mines, Louisiana in 1903.
When Frasch's patent expired, the process was widely applied to similar salt-dome sulfur deposits along the US Gulf Coast. The second Frasch-process mine opened in 1912 in Brazoria County, Texas. The US Gulf Coast came to dominate world sulfur production in the early and middle 20th century. However, starting in the 1970s, byproduct sulfur recovery from oil and natural gas lowered the price of sulfur and drove many Frasch-process mines out of business. The last US Frasch sulfur mine closed in 2000. A Frasch mine in Iraq closed in 2003 due to the U.S. invasion of Iraq.
The Frasch process is still used to work sulfur deposits in Mexico and Poland.
Process
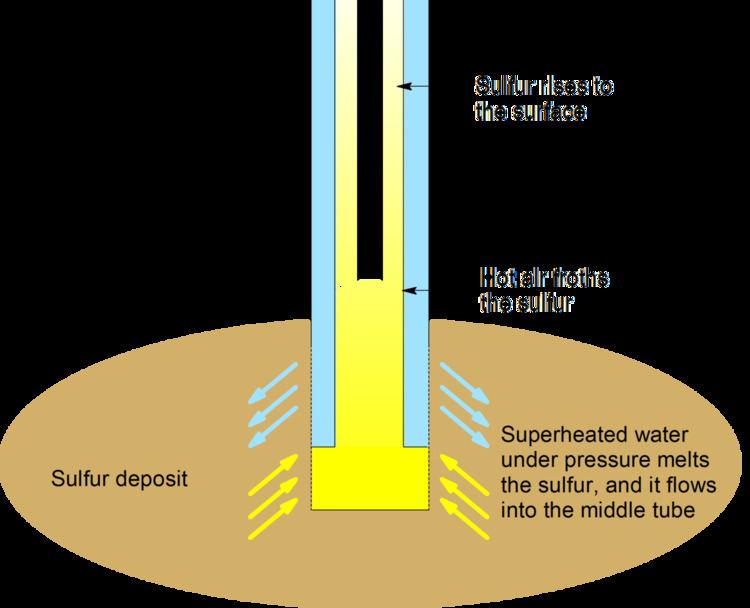
In the Frasch process, three concentric tubes are introduced into the sulfur deposit. Superheated water (443k,165 °C, 2.5-3 MPa) is injected into the deposit via the outermost tube. Sulfur (m.p. 115 °C) melts and flows into the middle tube. Water pressure alone is unable to force the sulfur into the surface due to the molten sulfur's greater density, so hot air is introduced via the innermost tube to froth the sulfur, making it less dense, and pushing it to the surface.
The sulfur obtained can be very pure (99.7 - 99.8%). In this form, it is light yellow in color. If contaminated by organic compounds, it can be dark-colored; further purification is not economic, and usually unnecessary. Using this method, the United States produced 3.89 million tons of sulfur in 1989, and Mexico produced 1.02 million tons of sulfur in 1991.
The Frasch process can be used for deposits 50–800 meters deep. 3-38 cubic meters of superheated water are required to produce every tonne of sulfur, and the associated energy cost is significant. A working demonstration model of the Frasch process suitable for the classroom has been described.
