Formula C13H10 Melting point 116 °C Boiling point 295 °C | Molar mass 166.223 g/mol Density 1.2 g/cm³ IUPAC ID 9H-Fluorene | |
 | ||
Fluorene
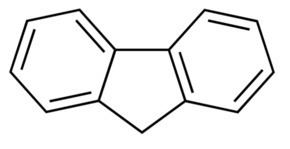
Fluorene /ˈflʊəriːn/, or 9H-fluorene, is a polycyclic aromatic hydrocarbon. It forms white crystals that exhibit a characteristic, aromatic odor similar to that of naphthalene. It is combustible. It has a violet fluorescence, hence its name. For commercial purposes it is obtained from coal tar. It is insoluble in water and soluble in many organic solvents.
Contents
Fluorene meaning
Synthesis, structure, and reactivity
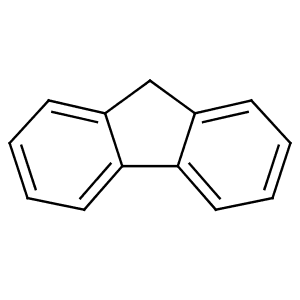
Although fluorene is obtained from coal tar, it can also be prepared by dehydrogenation of diphenylmethane. Alternatively, it can be prepared by the reduction of fluorenone with zinc. The fluorene molecule is nearly planar, although each of the two benzene rings is coplanar with the central carbon 9.
Acidity
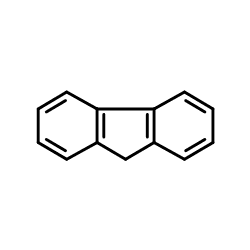
The C9-H sites of the fluorene ring are weakly acidic (pKa = 22.6 in DMSO.) Deprotonation gives the stable fluorenyl "anion", nominally C13H9−, which is aromatic and has an intense orange colour. The anion is a nucleophile, and most electrophiles react with it by adding to the 9-position. The purification of fluorene exploits its acidity and the low solubility of its sodium derivative in hydrocarbon solvents.
Both protons can be removed from C9. For example, 9,9-fluorenyldipotassium can be obtained by treating fluorene with potassium metal in boiling dioxane.
Uses
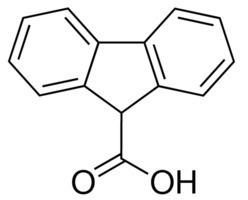
Fluorene is a precursor to other fluorene compounds; the parent species has few applications. Fluorene-9-carboxylic acid is a precursor to pharmaceuticals. 2-Aminofluorene, 3,6-bis-(dimethylamino)fluorene, and 2,7-diiodofluorene are precursors to dyes. Oxidation of fluorene gives fluorenone, which is nitrated to give commercially useful derivatives. 9-Fluorenylmethyl chloroformate (Fmoc chloride) is used to introduce the 9-fluorenylmethyl carbamate (Fmoc) protecting group on amines in peptide synthesis.
Polyfluorene polymers (where carbon 7 of one unit is linked to carbon 2 of the next one, displacing two hydrogens) are electrically conductive and electroluminescent, and have been much investigated as a luminophore in organic light-emitting diodes.
Cicloprofen is a NSAID 2-arylpropionic acid made from fluorene.
