Formula C9H10O2 Density 1.05 g/cm³ | Molar mass 150.17 g/mol Melting point -34 °C | |
 | ||
Related compounds | ||
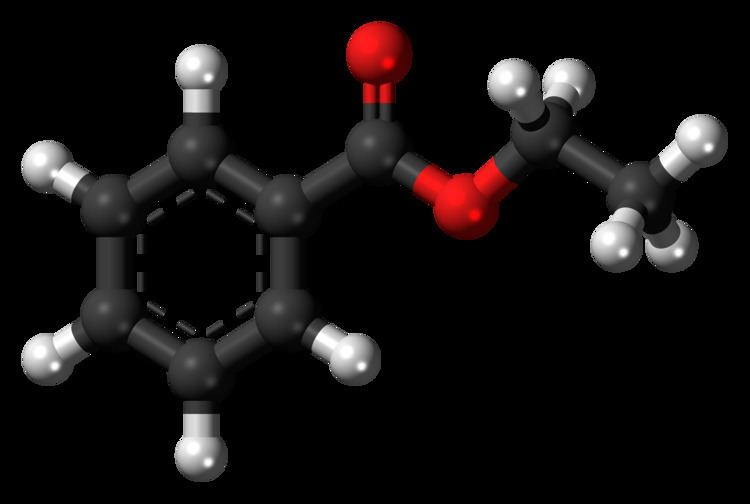
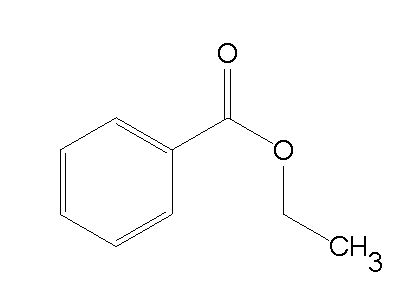
Ethyl benzoate, C9H10O2, is the ester formed by the condensation of benzoic acid and ethanol. It is a colorless liquid that is almost insoluble in water, but miscible with most organic solvents.
Contents

As with many volatile esters, ethyl benzoate has a pleasant odor described as sweet, wintergreen, fruity, medicinal, cherry, and grape. It is a component of some fragrances and artificial fruit flavors.
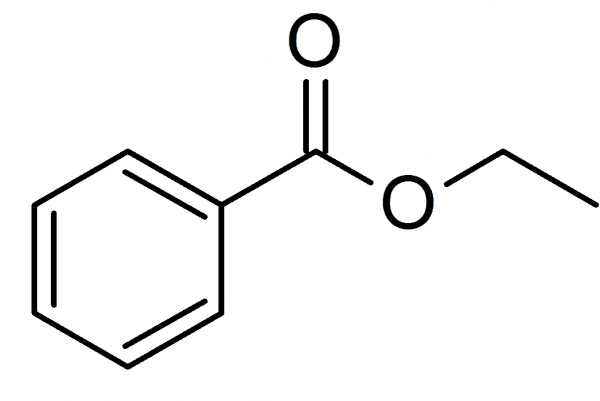
The fischer esterification methyl benzoate
Preparation

A simple and commonly used method for the preparation of ethyl benzoate in laboratory is the acidic esterification of benzoic acid with ethanol and sulfuric acid as catalyst:

References
Ethyl benzoate Wikipedia(Text) CC BY-SA
