Symbol EHHADH Entrez 1962 OMIM 607037 | Alt. symbols ECHD HUGO 3247 RefSeq NM_001966 | |
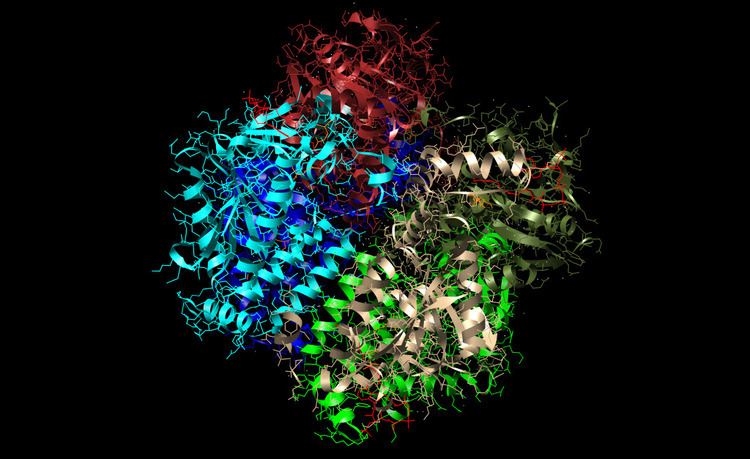 | ||
Enoyl-CoA hydratase is an enzyme that hydrates the double bond between the second and third carbons on acyl-CoA. This enzyme, also known as crotonase, is essential to metabolizing fatty acids to produce both acetyl CoA and energy. Note the crystal structure at right of enoyl-coa hydratase from a rat. The crystal structure shows a hexamer formation (not universal, but human enzyme is also hexameric), which leads to the efficiency of this protein. This enzyme has been discovered to be highly efficient, and allows our bodies to metabolize fatty acids into energy very quickly. In fact this enzyme is so efficient that the rate is equivalent to that of diffusion-controlled reactions.
Contents
Biological Significance: Metabolism
Enoyl-CoA hydratase catalyzes the second step in the breakdown of fatty acids or the second step of β-oxidation in fatty acid metabolism shown below. Fatty acid metabolism is how our bodies turn fats or lipids into energy. When fats come into our bodies, they are generally in the form of triacyl-glycerols. These must be broken down in order for the fats to pass into our bodies. When that happens, three fatty acids are released.
In fatty acid metabolism, fatty acids are changed into fatty acyl-CoA. To do this, the carboxylate which occupies one end of the fatty acid is changed into a thioester by substituting coenzyme A for the hydroxyl group. Next the fatty acyl-CoA is oxidized and broken down into an acetyl-CoA molecule and another acyl-CoA. The acetyl CoA is then sent to the citric acid cycle while the remaining acyl-CoA is broken down further into acetyl-CoAs. The complete breakdown of a fatty acid not only generates acetyl-CoA molecules, but it also generates energy in the form of NADH. This NADH goes on to be converted into ATP which can be used in other reactions.
Mechanism
Enoyl-CoA hydratase (ECH) is used in β-oxidation to add a hydroxyl group and a proton to the unsaturated β-carbon on a fatty-acyl CoA. The enzyme functions by providing two glutamate residues as catalytic acid and base. The two amino acids hold a water molecule in place, allowing it to attack in a syn addition to an α-β unsaturated acyl-CoA at the β-carbon. The α-carbon then grabs another proton, which completes the formation of the beta-hydroxy acyl-CoA.
It is also known from experimental data that no other sources of protons reside in the active site. This means that the proton which the α-carbon grabs is from the water that just attacked the β-carbon. What this implies is that the hydroxyl group and the proton from water are both added from the same side of the double bond, a syn addition. This allows the enzyme to make an S stereoisomer from 2-trans-enoyl-CoA and an R stereoisomer from the 2-cis-enoyl-CoA. This is made possible by the two glutamate residues which hold the water in position directly adjacent to the α-β unsaturated double bond, as seen in figure 1. This configuration requires that the active site for this enzyme is extremely rigid, to hold the water in a very specific configuration with regard to the acyl-CoA. The data for a mechanism for this reaction is not conclusive as to whether this reaction is concerted or occurs in consecutive steps. If occurring in consecutive steps, the intermediate is identical to that which would be generated from an E1cb elimination reaction. Both mechanisms are shown below.
The enzyme is mechanistically similar to fumarase.
It is classified as EC 4.2.1.17.
