Related alkanes Density 750 kg/m³ Appearance Colorless liquid | Boiling point 216.2 °C Formula C12H26 | |
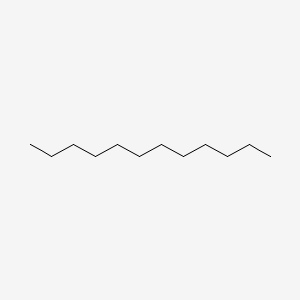 | ||
Dodecane (also known as dihexyl, bihexyl, adakane 12 or duodecane) is a liquid alkane hydrocarbon with the chemical formula CH3(CH2)10CH3 (or C12H26), an oily liquid of the paraffin series. It has 355 isomers.
Contents

It is used as a solvent, distillation chaser, and scintillator component. It is used as a diluent for tributyl phosphate (TBP) in reprocessing plants.
Combustion reaction
The combustion reaction of dodecane is as follows:
C12H26(l) + 18.5 O2(g) → 12 CO2(g) + 13 H2O(g)ΔH° = −7513 kJ
One litre of fuel needs about 15 kg of air to burn, and generates 2.3 kg (or 1.2 m3) of CO2 upon complete combustion.
Jet fuel surrogate

In recent years, n-dodecane has garnered attention as a possible surrogate for kerosene-based fuels such as Jet-A, S-8, and other conventional aviation fuels. It is considered a second-generation fuel surrogate designed to emulate the laminar flame speed, largely supplanting n-decane, primarily due to its higher molecular mass and lower hydrogen to carbon ratio which better reflect the n-alkane content of jet fuels.

