Related compounds Density 2.16 g/cm³ | Formula C4FeNa2O4 Appearance Colorless solid | |
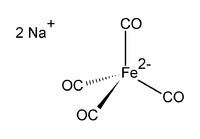 | ||
Disodium tetracarbonylferrate is the organometallic compound with the formula Na2[Fe(CO)4]. This oxygen-sensitive colourless solid is employed in organic synthesis, mainly to synthesise aldehydes. It is commonly used with dioxane complexed to the sodium cation, this dioxane solvate being known as Collman's reagent. The tetracarbonylferrate dianion is tetrahedral.
Contents
Synthesis
The reagent was reported by Cooke in 1970. The current synthesis entails the reduction of a solution of iron pentacarbonyl in tetrahydrofuran by sodium naphthenide. The efficiency of the synthesis depends on the quality of the iron pentacarbonyl.
Fe(CO)5 + 2 Na → Na2[Fe(CO)4] + COWhen a deficiency of sodium is used, the reduction affords octacarbonyl diferrate:
2 Fe(CO)5 + 2 Na → Na2[Fe2(CO)8] + 2 COOther synthesis forms to make Collman's Reagent from Fe(CO)5 are listed below.
Fe(CO)5 + Na-Hg +THF → Na2[Fe(CO)4]Fe(CO)5 + Na + Dioxane + PhCOPh → Na2[Fe(CO)4]Fe(CO)5 + Na/PhCOPh + THF → Na2[Fe(CO)4]Another way to synthesize Collman's Reagent is to use FeCl3.
FeCl3 + Na(C10H8) + 4CO + THF → +Na → Na2[Fe(CO)4]These synthesis pathways are extremely useful in preparing Collman's Reagent if the typical reagents to make it are not available.
Reactions
The reagent was originally described for the conversion of primary alkyl bromides, RBr, to the corresponding aldehydes in a two-step, "one-pot" reaction:
Na2[Fe(CO)4] + RBr → Na[RFe(CO)4] + NaBrThis solution is then treated sequentially with PPh3 and then acetic acid to give the aldehyde, RCHO.
Disodium tetracarbonylferrate can be used to convert acid chlorides to aldehydes. As for Cooke’s early discovery, an iron acyl complex undergoes protonolysis to give the aldehyde.
Na2[Fe(CO)4] + RCOCl → Na[RC(O)Fe(CO)4] + NaClNa[RC(O)Fe(CO)4] + HCl → RCHO + "Fe(CO)4" + NaClDisodium tetracarbonylferrate reacts with alkyl halides (RX) to produce alkyl complexes:
Na2[Fe(CO)4] + RX → Na[RFe(CO)4] + NaXSuch iron alkyls can be converted to the corresponding carboxylic acid and acid halides:
Na[RFe(CO)4] + O2, H+ →→ RCO2H + Fe...Na[RFe(CO)4] + 2 X2 → RC(O)X + FeX2 + 3 CO + NaX
One attraction of these methods is the low cost of the iron carbonyl as well as the fact that the procedures are relatively “green” because the side product is iron-based.
