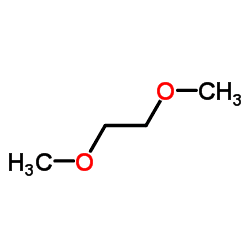
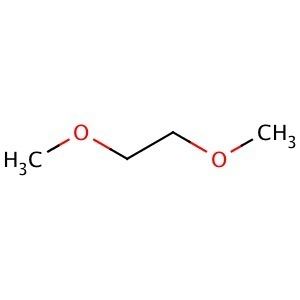
Boiling point 85 °C Density 868 kg/m³ Melting point -58 °C | Formula C4H10O2 Molar mass 90.12 g/mol Appearance Colorless liquid | |
 | ||
Related compounds | ||
Dimethoxyethane meaning
Dimethoxyethane, also known as glyme, monoglyme, dimethyl glycol, ethylene glycol dimethyl ether, dimethyl cellosolve, and DME, is a clear, colorless, aprotic, and liquid ether that is used as a solvent, especially in batteries. Dimethoxyethane is miscible with water.
Contents

Production
Monoglyme may be manufactured by a number of methods:

Applications as solvent and ligand
Together with a high-permittivity solvent (e.g. propylene carbonate), dimethoxyethane is used as the low-viscosity component of the solvent for electrolytes of lithium batteries. In the laboratory, DME is used as a coordinating solvent.

Dimethoxyethane is often used as a higher boiling alternative to diethyl ether and THF. Dimethoxyethane forms chelate complexes with cations and acts as a bidentate ligand. It is therefore often used in organometallic chemistry like Grignard reactions, hydride reductions, and palladium-catalyzed reactions like Suzuki reactions and Stille couplings. Dimethoxyethane is also a good solvent for oligo- and polysaccharides.