Density 1.58 g/cm³ | Boiling point 189.3 °C | |
 | ||
Related compounds Appearance Colorless or white crystals | ||
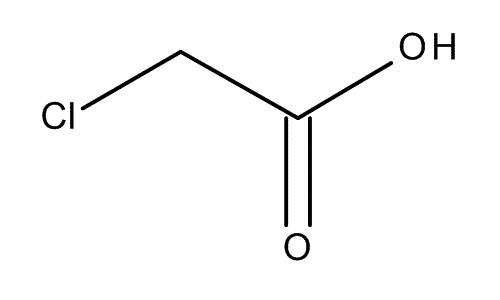
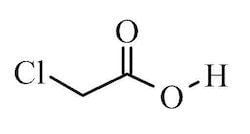
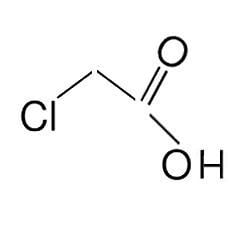
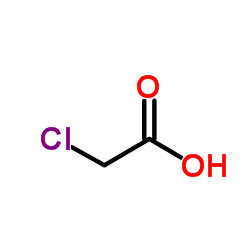
Chloroacetic acid, industrially known as monochloroacetic acid (MCA) is the organochlorine compound with the formula ClCH2CO2H. This carboxylic acid is a useful building-block in organic synthesis.
Contents
Synthesis of chloroacetic acid
History
Chloroacetic acid was first prepared (in impure form) by the French chemist Félix LeBlanc (1813–1886) in 1843 by chlorinating acetic acid in the presence of sunlight, and in 1857 (in pure form) by the German chemist Reinhold Hoffmann (1831–1919) by refluxing glacial acetic acid in the presence of chlorine and sunlight, and then by the French chemist Charles-Adolphe Wurtz by reacting chloroacetyl chloride (ClCH2COCl) with water, also in 1857.
Production
The production of chloroacetic acid was 706,000 tonnes/year in 2010, of which over half is produced in China. Other countries with significant production capacity are Germany (105,000), the Netherlands (100,000), India (>65,000), and the United States (55,000).
Processes
Chloroacetic acid is prepared industrially via two routes. The predominant method involves chlorination of acetic acid, with acetic anhydride as a catalyst.
CH3CO
2H + Cl
2 → ClCH
2CO
2H + HCl
The other main industrial route to chloroacetic acid is hydrolysis of trichloroethylene using sulfuric acid as a catalyst.
CCl2CHCl + 2 H
2O → ClCH
2CO
2H + 2 HCl
The hydrolysis method produces a highly pure product, which can be important since mono-, di-, and trichloroacetic acids are difficult to separate by distillation. Approximately, 420,000,000 kg/y are produced globally.
Reactivity and uses
Illustrative of its usefulness in organic chem. is the O-alkylation of salicylaldehyde with chloroacetic acid, followed by decarboxylation of the resulting ether, producing benzofuran.

In industry, chloroacetic acid is used in the production of a wide variety of useful compounds (e.g. drugs, dyes, and pesticides). Most reactions take advantage of the high reactivity of the C–Cl bond. It is the precursor to the herbicide glyphosate, and the herbicides MCPA (2-methyl-4-chlorophenoxyacetic acid) and dimethoate are prepared by alkylation with chloroacetic acid. Chloroacetic acid is converted to chloroacetyl chloride, a precursor to adrenaline (epinephrine). Displacement of chloride by sulfide gives thioglycolic acid, which is used as a stabilizer in PVC and a component in some cosmetics.
In its largest-scale application, chloroacetic acid is used to prepare the thickening agent carboxymethyl cellulose and carboxymethyl starch.
Safety
Like other chloroacetic acids and related halocarbons, chloroacetic acid is a hazardous alkylating agent. The LD50 for rats is 76 mg/kg.
Chloroacetic acid easily penetrates skin and mucous membranes and interferes with cellular energy production. Initial dermal exposure to high concentrations (e.g., 80% solution) may not appear very damaging at first, however systemic poisoning may present within hours. Exposure can be fatal if greater than 6% body surface area is exposed to chloroacetic acid. The sodium salt does not penetrate the skin as well as the acid but can be as damaging given a longer duration and greater surface area of exposure.
Upon exposure to chloroacetic acid, immediate decontamination should be commenced by rinsing the affected area with water or bicarbonate solution in order to neutralize the acid and prevent further skin absorption.
The antidote of chloroacetic acid poisoning is sodium dichloroacetate (50 mg/kg IV over 10 mins, repeated in 2h; double dosage if hemodialysis is performed).
It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.
