EC number 3.4.17.1 ExPASy NiceZyme view | CAS number 9031-98-5 | |
 | ||
Carboxypeptidase A usually refers to the pancreatic exopeptidase that hydrolyzes peptide bonds of C-terminal residues with aromatic or aliphatic side-chains. Most scientists in the field now refer to this enzyme as CPA1, and to a related pancreatic carboxypeptidase as CPA2.
In addition, there are 4 other mammalian enzymes named CPA-3 through CPA-6, and none of these are expressed in the pancreas. Instead, these other CPA-like enzymes have diverse functions.
CPA-1 and CPA-2 (and, it is presumed, all other CPAs) employ a zinc ion within the protein for hydrolysis of the peptide bond at the C-terminal end of an amino acid residue. Loss of the zinc leads to loss of activity, which can be replaced easily by zinc, and also by some other divalent metals (cobalt, nickel). Carboxypeptidase A is produced in the pancreas and is crucial to many processes in the human body to include digestion, post-translational modification of proteins, blood clotting, and reproduction. This vast scope of functionality for a single protein makes it the ideal model for research regarding other zinc proteases of unknown structure. Recent biomedical research on collagenase, enkephlinase, and angiotensin-converting enzyme used carboxypeptidase A for inhibitor synthesis and kinetic testing. For example, a drug that treats high blood pressure, Captopril, was designed based on a carboxypeptidase A inhibitor. Carboxypeptidase A and the target enzyme of Captopril, angiotensin-converting enzyme, have very similar structures, as they both contain a zinc ion within the active site. This allowed for a potent carboxypeptidase A inhibitor to be used to inhibit the enzyme and, thus, lower blood pressure through the renin-angiotensin-aldosterone system.
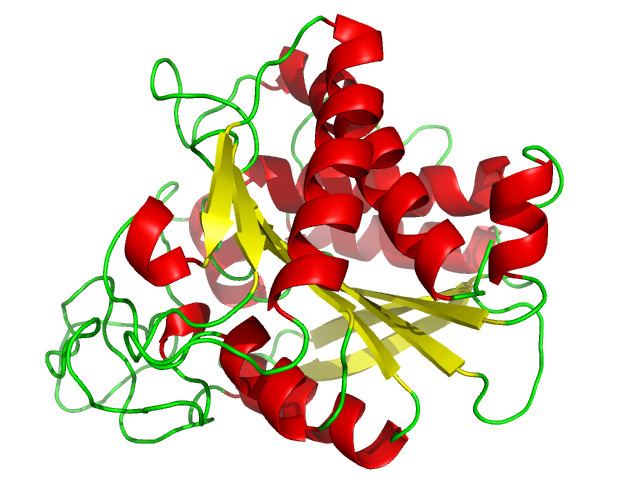
Classified as a metalloexopeptidase, carboxypeptidase A consists of a single polypeptide chain bound to a zinc ion. This characteristic metal ion is located within the active site of the enzyme, along with five amino acid residues that are involved in substrate binding: Arg-71, Arg-127, Asn-144, Arg-145, Tyr-248, and Glu-270. X-ray crystallographic studies have revealed five subsites on the protein. These allosteric sites are involved in creating the ligand-enzyme specificity seen in most bioactive enzymes. One of these subsites induces a conformational change at Tyr-248 upon binding of a substrate molecule at the primary active site. The phenolic hydroxyl of tyrosine forms a hydrogen bond with the terminal carboxylate of the ligand. In addition, a second hydrogen bond is formed between the tyrosine and a peptide linkage of longer peptide substrates. These changes make the bond between the enzyme and ligand, whether it is substrate or inhibitor, much stronger. This property of carboxypeptidase A led to the first clause of Daniel E. Koshland, Jr.’s “induced fit” hypothesis.
Several studies have been conducted exploring the details of the bond between carboxypeptidase A and substrate and how this affects the rate of hydrolysis. In 1934, it was first discovered through kinetic experiments that, in order for substrate to bind, the peptide that is to be hydrolyzed must be adjacent to a terminal free hydroxyl group. Also, the rate of hydrolysis can be enhanced if the C-terminal residue is branched aliphatic or aromatic. However, if the substrate is a dipeptide with a free amino group, it undergoes hydrolysis slowly; this, however, can be avoided if the amino group is blocked by N-acylation
It is quite clear that the structure of the enzyme, to be specific the active site, is very important in understanding the mechanism of reaction. For this reason, Rees and colleagues studied the enzyme-ligand complex to get a clear answer for the role of the zinc ion. These studies found that, in free enzyme, the zinc coordination number is five; the metal center is coordinated with two imidazole Nδ1 nitrogens, the two carboxylate oxygens of glutamate-72, and a water molecule to form a distorted tetrahedral. However, once ligand binds at the active site of carboxypeptidase A, this coordination number can vary from five to six. When bound to dipeptide glycyl-L-tyrosine, the amino nitrogen of the dipeptide and the carbonyl oxygen replaced the water ligand. This would yield a coordination number of six for the zinc in the carboxypeptidase A- dipeptide glycyl-L-tyrosine complex. Electron density maps gave evidence that the amino nitrogen occupies a second position near glutamate-270. The closeness of these two residues would result in a steric hindrance preventing the water ligand from coordinating with zinc. This would result in a coordination number of five. Data for both are substantial, indicating that both situations occur naturally
There are two proposed mechanisms for the catalytic function of carboxypeptidase A. The first is a nucleophilic pathway involving a covalent acyl enzyme intermediate containing active site base Glu-270. Evidence for this anhydride intermediate is mixed; Suh and colleagues isolated what is assumed to by the acyl intermediate. However, confirmation of the acyl enzyme was done without trapping experiments, making the conclusions weak.
The second proposed mechanism is a promoted water pathway. This mechanism involves attack of a water molecule at the scissile peptide linkage of the substrate. This process is promoted by the zinc ion and assisted by residue Glu-270
