 | ||
Carboxylation in chemistry is a chemical reaction in which a carboxylic acid group is introduced in a substrate. The opposite reaction is decarboxylation.
Contents
Carboxylation in organic chemistry
In organic chemistry many different protocols exist for carboxylation. One general approach is by reaction of nucleophiles with dry ice (solid carbon dioxide) or formic acid An example is the Kolbe–Schmitt reaction. Carboxylation catalysts are N-Heterocyclic carbenes and catalysts based on silver.
Carboxylation in biochemistry
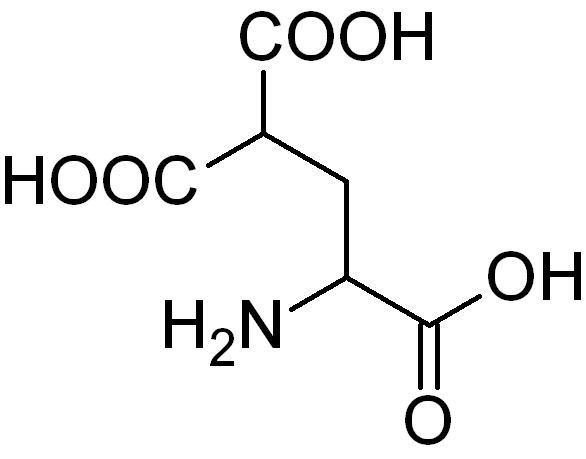
Carboxylation in biochemistry is a posttranslational modification of glutamate residues, to γ-carboxyglutamate, in proteins. It occurs primarily in proteins involved in the blood clotting cascade, specifically factors II, VII, IX, and X, protein C, and protein S, and also in some bone proteins. This modification is required for these proteins to function. Carboxylation occurs in the liver and is performed by γ-glutamyl carboxylase.
The carboxylase requires vitamin K as a cofactor and performs the reaction in a processive manner. γ-carboxyglutamate binds calcium, which is essential for its activity. For example, in prothrombin, calcium binding allows the protein to associate with the plasma membrane in platelets, bringing it into close proximity with the proteins that cleave prothrombin to active thrombin after injury.
