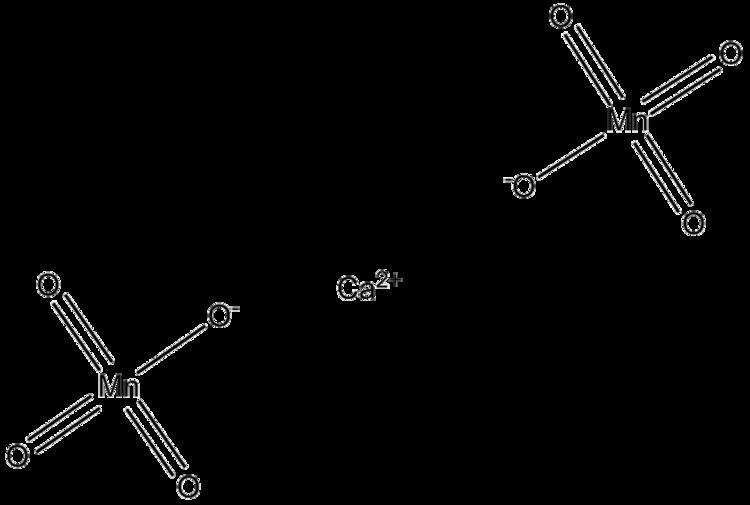
Formula Ca(MnO4)2 Density 2.49 g/cm³ | Molar mass 277.949 g/mol Melting point 140 °C | |
 | ||
Appearance purple crystals; deliquesent | ||
Calcium permanganate is an oxidizing agent and chemical compound with the chemical formula Ca(MnO4)2. It consists of the metal calcium and two permanganate ions. It is noncombustible, but, being a strong oxidizing agent, it will accelerate the burning of combustible material. If the combustible material is finely divided, the resulting mixture may be explosive. Contact with liquid combustible materials may result in spontaneous ignition. Contact with sulfuric acid may cause fires or explosions. Mixtures with acetic acid or acetic anhydride can explode if not kept cold. Explosions can occur when mixtures of calcium permanganate and sulfuric acid come into contact with benzene, carbon disulfide, diethyl ether, ethyl alcohol, petroleum, or other organic matter.
It is prepared from the reaction of potassium permanganate with calcium chloride or from the reaction of aluminum permanganate with calcium oxide.
Uses
It is believed to help whiten teeth.
