Formula CsBr Density 4.44 g/cm³ Boiling point 1,300 °C | Molar mass 212.81 g/mol Melting point 636 °C Appearance White solid | |
 | ||
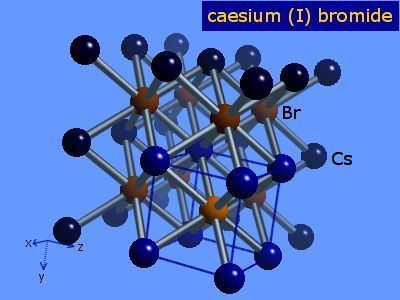
Caesium bromide, (CsBr), is an ionic compound of caesium and bromine. It has simple cubic p-type cubic crystallic structure, comparable to that of caesium chloride type with space group Pm3m and lattice constant a = 0.42953 nm. The distance between Cs+ and Br− ions is 0.37198 nm.
Contents
Synthesis
It can be prepared via following reactions:

The direct synthesis is a vigorous reaction of caesium with other halogens. Due to its high cost, it is not used for preparation.
Uses
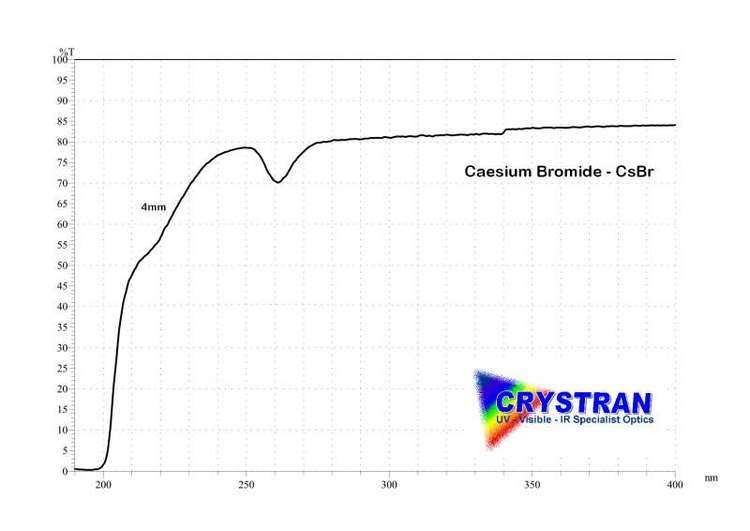
Caesium bromide is sometimes used in optics as a beamsplitter component in wide-band spectrophotometers.


References
Caesium bromide Wikipedia(Text) CC BY-SA
