Molar mass 230.5 g/mol Density 2.34 g/cm³ | Melting point 255 °C | |
 | ||
Appearance colorless crystals (anhydrous)white crystals (dihydrate) | ||
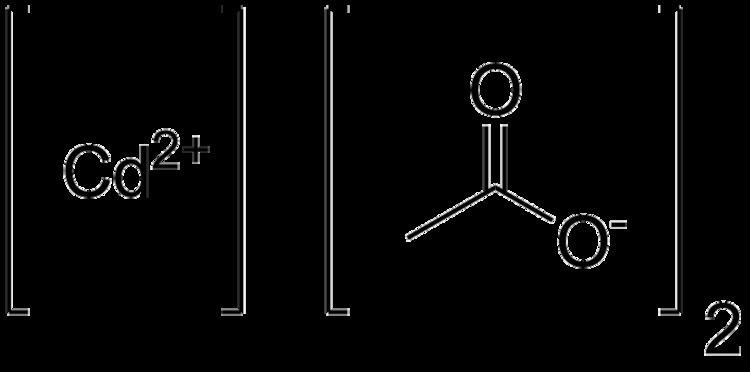
Cadmium acetate is the chemical compound with the formula Cd(CH3CO2)2. This colourless solid is classified coordination polymer, featuring acetate ligands interconnecting cadmium centers. The compound exists in anhydrous form and as a dihydrate. It forms by treating cadmium oxide with acetic acid:
Contents
CdO + 2 CH3COOH → Cd(CH3COO)2 + H2OUses
Cadmium acetate is used for glazing ceramics and pottery; in electroplating baths, in dyeing and printing textiles; and as an analytic reagent for sulfur, selenium and tellurium.
Preparation
Cadmium acetate is prepared by treating cadmium oxide with acetic acid. The compound may also be prepared by treating cadmium nitrate with acetic anhydride.
Safety
Cadmium compounds are considered Group 1 carcinogens by the IARC.
References
Cadmium acetate Wikipedia(Text) CC BY-SA
