Abbreviations R-20B3UN 2515 Formula CHBr3 Density 2.89 g/cm³ | Appearance Colorless liquid Boiling point 149.1 °C Melting point 8 °C | |
 | ||
Related alkanes Thermodynamicdata Phase behavioursolid–liquid–gas | ||
Synthesis of bromoform revisit of iodoform
Bromoform (CHBr3) is a brominated organic solvent, colorless liquid at room temperature, with a high refractive index, very high density, and sweet odor is similar to that of chloroform. It is a trihalomethane, and is one of the four haloforms, the others being fluoroform, chloroform, and iodoform. Bromoform can be prepared by the haloform reaction using acetone and sodium hypobromite, by the electrolysis of potassium bromide in ethanol, or by treating chloroform with aluminum bromide. Currently its main use is as a laboratory reagent.
Contents
- Synthesis of bromoform revisit of iodoform
- Synthesis of bromoform
- Structure
- Uses
- Environment and Toxicology
- References

Synthesis of bromoform
Structure
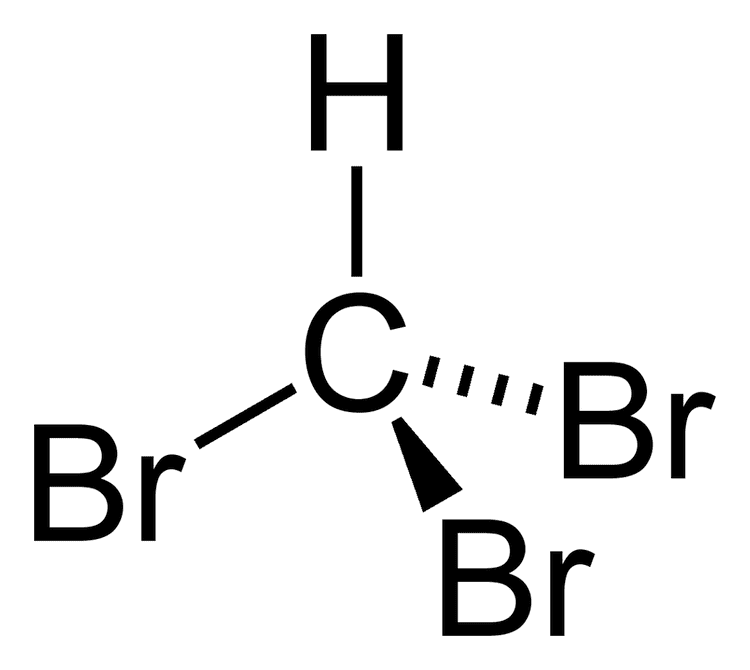
The molecule adopts tetrahedral molecular geometry with C3v symmetry.
Uses
Only small quantities of bromoform are currently produced industrially in the United States. In the past, it was used as a solvent, sedative and flame retardant, but now it is mainly used as a laboratory reagent, for example as an extraction solvent.

Bromoform's high density makes it useful for separation of minerals by density. When two samples are mixed with bromoform and then allowed to settle, the top layer will contain minerals lighter than bromoform, and the bottom layer will contain heavier minerals. Slightly less dense minerals can be separated in the same way by mixing the bromoform with a small amount of a less dense and fully miscible solvent.
Environment and Toxicology

Natural production of bromoform by phytoplankton and seaweeds in the ocean is thought to be its predominant source in the environment. However, locally significant amounts of bromoform enter the environment formed as disinfection byproducts known as trihalomethanes when chlorine is added to drinking water to kill bacteria. It is somewhat soluble in water and readily evaporates into the air. Bromoform is the main trihalomethane produced in salt swimming pools with some public swimming pools found to contain up to 1.3 ppm (parts per million) bromoform. Occupational skin exposure limits are set at 0.5 ppm.

The substance may be hazardous to the environment, and special attention should be given to aquatic organisms. Its volatility and environmental persistence makes bromoform's release, either as liquid or vapor, strongly inadvisable.

Bromoform can be absorbed into the body by inhalation and through the skin. The substance is irritating to the respiratory tract, the eyes, and the skin, and may cause effects on the central nervous system and liver, resulting in impaired functions. It is soluble in about 800 parts water and is miscible with alcohol, benzene, chloroform, ether, petroleum ether, acetone, and oils. Its LD50 is 7.2 mmol/kg in mice, or 1.8g/kg. Bromoform is a confirmed animal carcinogen; (ACGIH 2004). Carcinogen category: 3B; (DFG 2004).
