In quantum physics and quantum chemistry, an avoided crossing (sometimes called intended crossing, non-crossing or anticrossing) is the phenomenon where two eigenvalues of an Hermitian matrix representing a quantum observable and depending on N continuous real parameters cannot become equal in value ("cross") except on a manifold of N-2 dimensions. In the case of a diatomic molecule (with one parameter, namely the bond length), this means that the eigenvalues cannot cross at all. In the case of a triatomic molecule, this means that the eigenvalues can coincide only at a single point (see conical intersection).
This is particularly important in quantum chemistry. In the Born–Oppenheimer approximation, the electronic molecular Hamiltonian is diagonalized on a set of distinct molecular geometries (the obtained eigenvalues are the values of the adiabatic potential energy surfaces). The geometries for which the potential energy surfaces are avoiding to cross are the locus where the Born–Oppenheimer approximation fails.
Study of a two-level system is of vital importance in quantum mechanics because it embodies simplification of many of physically realizable systems. The effect of perturbation on a two-state system Hamiltonian is manifested through avoided crossings in the plot of individual energy vs energy difference curve of the eigenstates. The two-state Hamiltonian can be written as
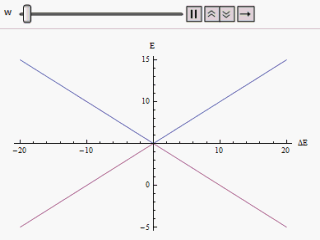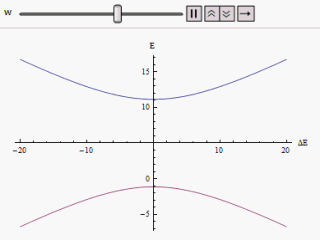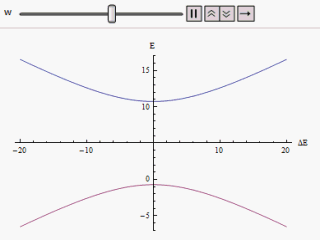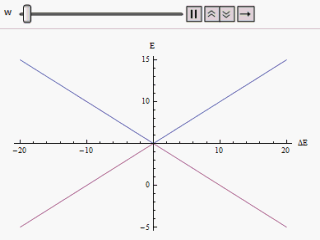
H = ( E 1 0 0 E 2 ) The eigenvalues of which are E 1 and E 2 and the eigenvectors, ( 1 0 ) and ( 0 1 ) . These two eigenvectors designate the two states of the system. If the system is prepared in either of the states it would remain in that state. If E 1 happens to be equal to E 2 there will be a twofold degeneracy in the Hamiltonian. In that case any mixed state of the degenerate eigenstates is evidently another eigenstate of the Hamiltonian. Hence the system prepared in any state will remain in that forever.
However, when subjected to an external perturbation, the matrix elements of the Hamiltonian change. For the sake of simplicity we consider a perturbation with only off diagonal elements. Since the overall Hamiltonian must be Hermitian we may simply write the new Hamiltonian
H ′ = H + P = ( E 1 0 0 E 2 ) + ( 0 W W ∗ 0 ) = ( E 1 W W ∗ E 2 ) Where P is the perturbation with zero diagonal terms. The fact that P is Hermitian fixes its off-diagonal components. The modified eigenstates can be found by diagonalising the modified Hamiltonian. It turns out that the new eigenvalues are,
E + = 1 2 ( E 1 + E 2 ) + 1 2 ( E 1 − E 2 ) 2 + 4 | W | 2 E − = 1 2 ( E 1 + E 2 ) − 1 2 ( E 1 − E 2 ) 2 + 4 | W | 2 If a graph is plotted varying ( E 1 − E 2 ) as abscissa and E + or E − as ordinate we find two branches of a hyperbola (as shown in the figure). The curve asymptotically approaches the original unperturbed energy levels. Analyzing the curves it becomes evident that even if the original states were degenerate (i.e. E 1 = E 2 ) the new energy states are no longer equal. However, if W is set to zero we may find at ( E 1 − E 2 ) = 0 , E + = E − and the levels cross. Thus with the effect of the perturbation these level crossings are avoided.
Avoided crossing and quantum resonance
The immediate impact of avoided level crossing in a degenerate two state system is the emergence of a lowered energy eigenstate. The effective lowering of energy always correspond to increasing stability. Bond resonance in organic molecules exemplifies the occurrence of such avoided crossings. To describe these cases we may note that the non-diagonal elements in an erstwhile diagonalised Hamiltonian not only modify the energy eigenvalues but also mix the old eigenstates into the new ones. These effects are more prominent if the original Hamiltonian had degeneracy. This mixing of eigenstates to attain more stability is precisely the phenomena of chemical bond resonance.
Our earlier treatment started by denoting the eigenvectors ( 1 0 ) and ( 0 1 ) as the matrix representation of eigenstates | ψ 1 ⟩ and | ψ 2 ⟩ of a two-state system. Using bra–ket notation the matrix elements of H ′ are actually the terms
H i j ′ = ⟨ ψ i | H ′ | ψ j ⟩ with
i , j ∈ { 1 , 2 } where H 11 ′ = H 22 ′ = E due to the degeneracy of the unperturbed Hamiltonian and the off-diagonal perturbations are H 12 ′ = W and H 21 ′ = W ∗ .
The new eigenstates | ψ + ⟩ and | ψ − ⟩ can be found by solving the eigenvalue equations H ′ | ψ + ⟩ = E + | ψ + ⟩ and H ′ | ψ − ⟩ = E − | ψ − ⟩ . From simple calculations it can be shown that
| ψ + ⟩ = 1 2 ( e i ϕ 1 ) = 1 2 ( e i ϕ | ψ 1 ⟩ + | ψ 2 ⟩ ) and
| ψ − ⟩ = 1 2 ( − e i ϕ 1 ) = 1 2 ( − e i ϕ | ψ 1 ⟩ + | ψ 2 ⟩ ) where
e i ϕ = W / | W | It is evident that both of the new eigenstates are mixture of the original degenerate eigenstates and one of the eigenvalues (here E − ) is less than the original unperturbed eigenenergy. So the corresponding stable system will naturally mix up the former unperturbed eigenstates to minimize its energy. In the example of Benzene the experimental evidences of probable bond structures give rise to two different eigenstates, | ψ 1 ⟩ and | ψ 2 ⟩ . The symmetry of these two structures mandates that ⟨ ψ 1 | H | ψ 1 ⟩ = ⟨ ψ 2 | H | ψ 2 ⟩ = E .
However it turns out that the two-state Hamiltonian H of Benzene is not diagonal. The off-diagonal elements result into lowering of energy and the Benzene molecule stabilizes in a structure which is a superposition of these symmetric ones with energy E − < E . For any general two-state system avoided level crossing repels the eigenstates | ψ + ⟩ and | ψ − ⟩ such that it requires more energy for the system to achieve the higher energy configuration.
The above illustration of avoided crossing however is a very specific case. From a generalised view point the phenomenon of avoided crossing is actually controlled by the parameters behind the perturbation. For the most general perturbation P = ( W 1 W W W 2 ) affecting a two-dimensional subspace of the Hamiltonian H we may write the effective Hamiltonian matrix in that subspace as,
( E 1 0 0 E 2 ) + ( W 1 W W W 2 ) = ( E 1 + W 1 W W E 2 + W 2 ) Here the elements of the state vectors were chosen to be real so that all the matrix elements become real. Now the eigenvalues of the system for this subspace is given by
E ± = 1 2 ( E 1 + E 2 + W 1 + W 2 ) ± 1 2 ( E 1 − E 2 + W 1 − W 2 ) 2 + 4 W 2 The terms under the square root are squared real numbers. So for these two levels to cross we must simultaneously require
( E 1 − E 2 + W 1 − W 2 ) = 0 W = 0 Now if the perturbation P has k parameters α 1 , α 2 , α 3 . . . . . α k we may in general vary these numbers to satisfy these two equations.
( E 1 − E 2 + W 1 − W 2 ) = F 1 ( α 1 , α 2 , α 3 . . . . . α k ) = 0 W = F 2 ( α 1 , α 2 , α 3 . . . . . α k ) = 0 If we choose the values of α 1 to α k − 1 then both of the equations above has one single free parameter. In general it is not possible to find one α k such that both of the equations are satisfied. However, if we allow another parameter to be free both of these two equations will now be controlled by the same two parameters
F 1 ( α k − 1 , α k ) | α 1 , α 2 , . . . , α k − 2 f i x e d = 0 F 2 ( α k − 1 , α k ) | α 1 , α 2 , . . . , α k − 2 f i x e d = 0 And generally there will be two such values of them for which the equations will simultaneously satisfy. So with k distinct parameters k − 2 parameters can always be chosen arbitrarily and still we can find two such α k 's such that there would be crossing of energy eigenvalues. In other words, the values of E + and E − would be the same for k − 2 freely varying co-ordinates(While the rest of the two co-ordinates are fixed from the condition equations). Geometrically the eigenvalue equations describe a surface in k dimensional space.
E ± = E ± ( α 1 , α 2 , α 3 . . . . . α k ) Since their intersection is parametrized by k − 2 coordinates we may formally state that for k continuous real parameters controlling the perturbed Hamiltonian, the levels(or surfaces) can only cross at a manifold of k − 2 dimension. However the symmetry of the Hamiltonian has a role to play in the dimensionality. If the original Hamiltonian has asymmetric states, ⟨ ψ 1 | P | ψ 2 ⟩ ≠ ⟨ ψ 2 | P | ψ 1 ⟩ , the off-diagonal terms vanish automatically to ensure hermiticity. This allows us to get rid of the equation W = 0 . Now from similar arguments as posed above it is straightforward that for asymmetrical Hamiltonian the intersection of energy surfaces takes place in a manifold of k − 1 dimension.
In polyatomic molecules, there are various parameters which determine the Hamiltonian of the system. The mutual distances between the atoms are one set of parameters. If both of the atoms of a diatomic molecule is same, the symmetry suggests that different configurations keeping their mutual distance fixed will result into same electronic states. So it is the relative distance r which acts as a parameter for the two equations promising level crossing. Hence due to the avoided crossing theorem in general we can not have level crossings between two electronic states of same symmetry. But in polyatomic molecules the number of independent mutual distances of nuclei are more. For a N-atomic molecule the number of independent mutual separation is S = 3 N − 6 (for N ≥ 3 ). Each of them acts as a parameter for the total Hamiltonian. Since we always have minimum of three independent parameters, level crossing is not totally avoided in these molecules.