Appearance colorless gas Molar mass 169.9136 g/mol Density 2.14 g/cm³ | Formula AsF5 Boiling point -52.8 °C | |
 | ||
Related compounds | ||
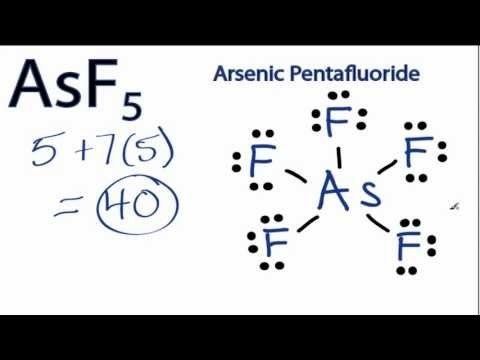
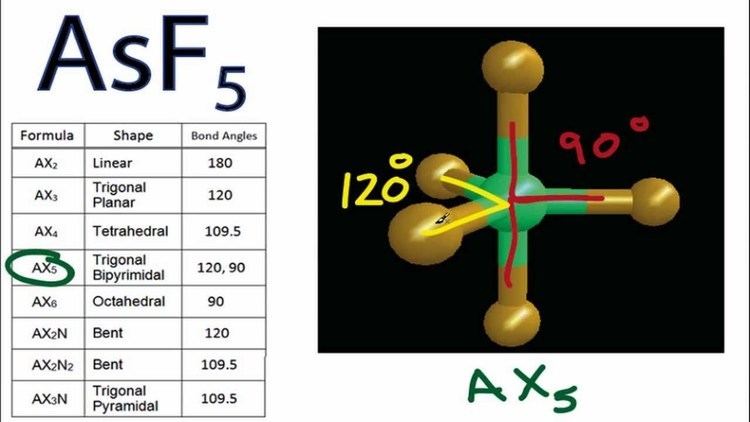
Asf5 molecular geometry and bond angles arsenic pentafluoride
Arsenic pentafluoride is a chemical compound of arsenic and fluorine. The oxidation state of arsenic is +5.
Contents
- Asf5 molecular geometry and bond angles arsenic pentafluoride
- Synthesis
- Properties
- Reactions
- Safety
- References
Synthesis
Arsenic pentafluoride can be prepared by direct combination of arsenic and fluorine:
2As + 5F2 → 2AsF5It can also be prepared by the reaction of arsenic trifluoride and fluorine:
AsF3 + F2 → AsF5or the addition of fluorine to arsenic pentoxide or arsenic trioxide.
2As2O5 + 10F2 → 4AsF5 + 5O22As2O3 + 10F2 → 4AsF5 + 3O2Properties
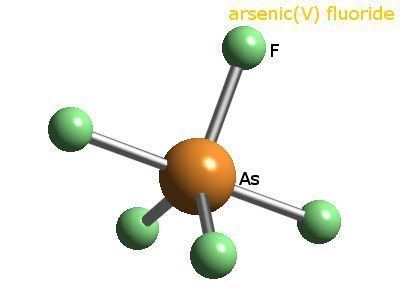
Arsenic pentafluoride is a colourless gas and has a trigonal bipyramidal structure. In the solid state the axial As-F bond lengths are 171.9 pm and the equatorial 166.8 pm.
Reactions

Arsenic pentafluoride forms halide complexes and is a powerful acceptor as shown by the reaction with sulfur tetrafluoride forming an ionic complex.
AsF5 + SF4 → SF3+ + AsF6−Safety
Arsenic pentafluoride is an extremely dangerous toxin, mainly poisoning liver cells. It has a smell that is similar to vinyl chloride gas.