 | ||
Antigenic shift is the process by which two or more different strains of a virus, or strains of two or more different viruses, combine to form a new subtype having a mixture of the surface antigens of the two or more original strains. The term is often applied specifically to influenza, as that is the best-known example, but the process is also known to occur with other viruses, such as visna virus in sheep. Antigenic shift is a specific case of reassortment or viral shift that confers a phenotypic change.
Contents
- Role in transmission of influenza viruses from non human animals to people
- Segmentation
- In the marine ecosystem
- References
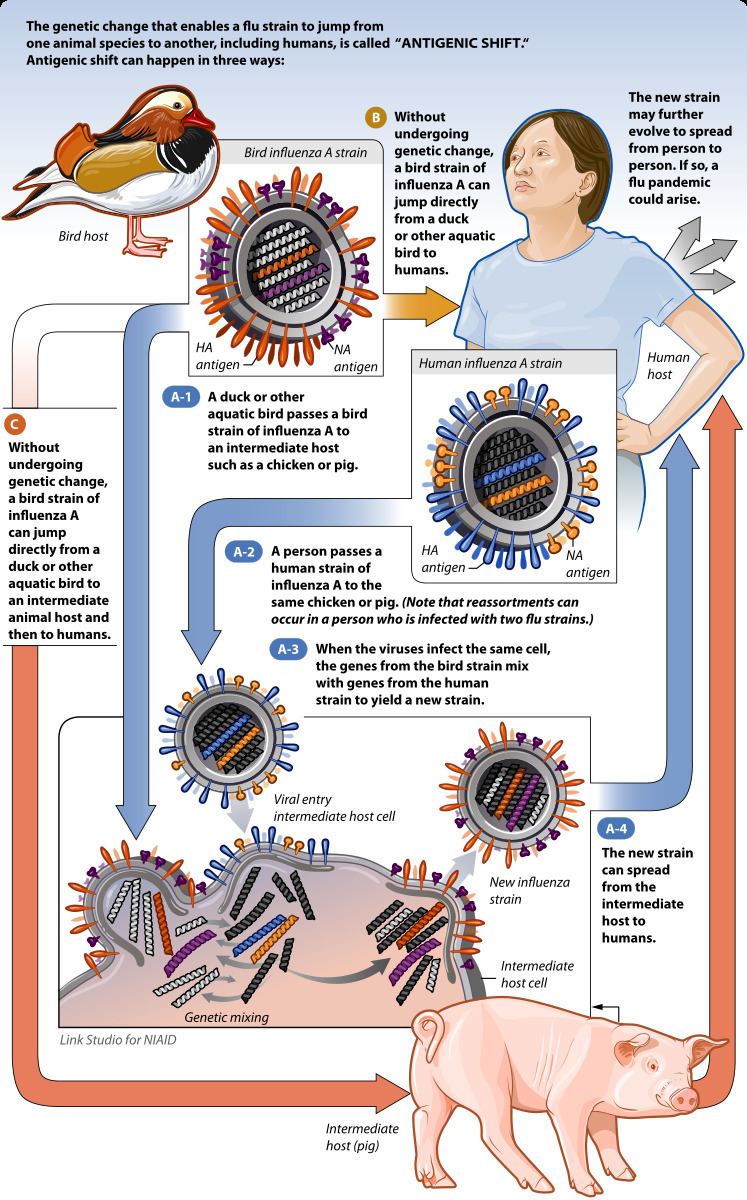
Antigenic shift is contrasted with antigenic drift, which is the natural mutation over time of known strains of influenza (or other things, in a more general sense) which may lead to a loss of immunity, or in vaccine mismatch. Antigenic drift occurs in all types of influenza including influenzavirus A, influenza B and influenza C. Antigenic shift, however, occurs only in influenzavirus A because it infects more than just humans. Affected species include other mammals and birds, giving influenza A the opportunity for a major reorganization of surface antigens. Influenza B and C principally infect humans, minimizing the chance that a reassortment will change its phenotype drastically.
Antigenic shift is important for the emergence of new viral pathogens as it is a pathway that viruses may follow to enter a new niche. It could occur with primate viruses and may be a factor for the appearance of new viruses in the human species such as HIV. Due to the structure of its genome, HIV does not undergo reassortment, but it does recombine freely and via superinfection HIV can produce recombinant HIV strains that differ significantly from their ancestors.
Role in transmission of influenza viruses from non-human animals to people
Influenza A viruses are found in many different animals, including ducks, chickens, pigs, humans, whales, horses, and seals. Influenza B viruses circulate widely principally among humans, though it has recently been found in seals. Flu strains are named after their types of hemagglutinin and neuraminidase surface proteins (of which there are 18 and 9 respectively), so they will be called, for example, H3N2 for type-3 hemagglutinin and type-2 neuraminidase. Some strains of avian influenza (from which all other strains of influenza A are believed to stem) can infect pigs or other mammalian hosts. When two different strains of influenza infect the same cell simultaneously, their protein capsids and lipid envelopes are removed, exposing their RNA, which is then transcribed to mRNA. The host cell then forms new viruses that combine their antigens; for example, H3N2 and H5N1 can form H5N2 this way. Because the human immune system has difficulty recognizing the new influenza strain, it may be highly dangerous, and result in a new pandemic.
Influenza viruses which have undergone antigenic shift have caused the Asian Flu pandemic of 1957, the Hong Kong Flu pandemic of 1968, and the Swine Flu scare of 1976. Until recently, such combinations were believed to have caused the infamous Spanish Flu outbreak of 1918 which killed 40~100 million people worldwide; however more recent research suggests the 1918 pandemic was caused by the antigenic drift of a fully avian virus to a form that could infect humans efficiently. The most recent 2009 H1N1 outbreak was a result of antigenic shift and reassortment between human, avian, and swine viruses. One increasingly worrying situation is the possible antigenic shift between avian influenza and human influenza. This antigenic shift could cause the formation of a highly virulent virus.
Segmentation
Antigenic shift occurs because the genome of the virus is segmented, allowing for dramatic genetic changes of type by re-assortment of its segmented RNA genome. A mnemonic to remember segmented viruses is BOAR: Bunyaviridae, Orthomyxoviridae, Arenavirus and Reoviridae.
In the marine ecosystem
In terms of virology, the marine ecosystem has been largely unstudied, but due to its extraordinary volume, high viral density (100 million viruses per mL in coastal waters, 3 million per mL in the deep sea) and high cell lysing rate (as high as 20% on average), marine viruses' antigenic shift and genetic recombination rates must be quite high. This is most striking when one considers that the coevolution of prokaryotes and viruses in the aquatic environment has been going on since before eukaryotes appeared on earth.
