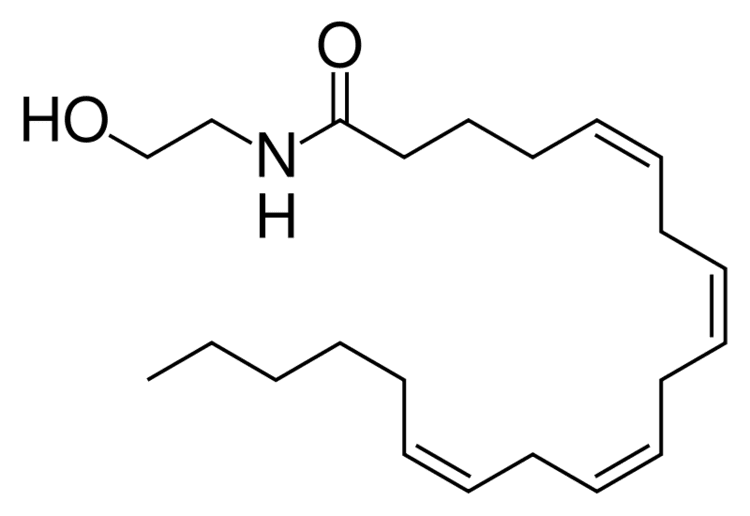
Formula C22H37NO2 Boiling point 522.3 °C | Molar mass 347.53 g/mol | |
 | ||
Cannabis marijuana thc anandamide
Anandamide, also known as N-arachidonoylethanolamine or AEA, is a fatty acid neurotransmitter derived from the non-oxidative metabolism of eicosatetraenoic acid (arachidonic acid) an essential ω-6 polyunsaturated fatty acid. The name is taken from the Sanskrit word ananda, which means "joy, bliss, delight", and amide. It is synthesized from N-arachidonoyl phosphatidylethanolamine by multiple pathways. It is degraded primarily by the fatty acid amide hydrolase (FAAH) enzyme, which converts anandamide into ethanolamine and arachidonic acid. As such, inhibitors of FAAH lead to elevated anandamide levels and are being pursued for therapeutic use.
Contents
- Cannabis marijuana thc anandamide
- D u a l i t y by anandamide feat soumalya rahul
- Physiological functions
- Synthesis and degradation
- Medical benefits
- References
D u a l i t y by anandamide feat soumalya rahul
Physiological functions
Anandamide was first described in 1992 by W. A. Devane and Lumír Hanuš.
Anandamide's effects can occur in either the central or peripheral nervous system. These distinct effects are mediated primarily by CB1 cannabinoid receptors in the central nervous system, and CB2 cannabinoid receptors in the periphery. The latter are mainly involved in functions of the immune system. Cannabinoid receptors were originally discovered as being sensitive to Δ9-tetrahydrocannabinol (Δ9-THC, commonly called THC), which is the primary psychoactive cannabinoid found in cannabis. The discovery of anandamide came from research into CB1 and CB2, as it was inevitable that a naturally occurring (endogenous) chemical would be found to affect these receptors.
Anandamide has been shown to impair working memory in rats. Studies are under way to explore what role anandamide plays in human behavior, such as eating and sleep patterns, and pain relief.
Anandamide is also important for implantation of the early stage embryo in its blastocyst form into the uterus. Therefore, cannabinoids such as Δ9-THC might influence processes during the earliest stages of human pregnancy. Peak plasma anandamide occurs at ovulation and positively correlates with peak estradiol and gonadotrophin levels, suggesting that these may be involved in the regulation of AEA (anandamide) levels. Subsequently, anandamide has been proposed as a biomarker of infertility, but so far lacks any predictive values in order to be used clinically.
Anandamide plays a role in the regulation of feeding behavior, and the neural generation of motivation and pleasure. In addition, anandamide injected directly into the forebrain reward-related brain structure nucleus accumbens enhances the pleasurable responses of rats to a rewarding sucrose taste, and enhances food intake as well. Moreover, the acute beneficial effects of exercise (termed as runner's high) seem to be mediated by anandamide in mice.
Anandamide is the precursor of a class of physiologically active substances, the prostamides.
Anandamide inhibits human breast cancer cell proliferation.
Anandamide is found in chocolate together with two substances that might mimic the effects of anandamide, N-oleoylethanolamine and N-linoleoylethanolamine.
Additionally, anandamide and other endocannabinoids are found in the model organism Drosophila melanogaster (fruit fly).
Synthesis and degradation
In humans, anandamide is biosynthesized from N-arachidonoyl phosphatidylethanolamine (NAPE). In turn NAPE arises by transfer of arachidonic acid from lecithin to the free amine of cephalin through an N-acyltransferase enzyme. Anandamide synthesis from NAPE occurs via multiple pathways and includes enzymes such as phospholipase A2, phospholipase C and NAPE-PLD.
The crystal structure of NAPE-PLD in complex with phosphatidylethanolamine and deoxycholate shows how the cannabinoid anandamide is generated from membrane N-acylphosphatidylethanolamines (NAPEs), and reveals that bile acids - which are mainly involved in the adsorption of lipids in the small intestine - modulate its biogenesis.
Endogenous anandamide is present at very low levels and has a very short half-life due to the action of the enzyme fatty acid amide hydrolase (FAAH), which breaks it down into free arachidonic acid and ethanolamine. Studies of piglets show that dietary levels of arachidonic acid and other essential fatty acids affect the levels of anandamide and other endocannabinoids in the brain. High fat diet feeding in mice increases levels of anandamide in the liver and increases lipogenesis. This suggests that anandamide may play a role in the development of obesity, at least in rodents.
Paracetamol (or acetaminophen in the U.S.A.) is metabolically combined with arachidonic acid by FAAH to form AM404. This metabolite of paracetamol is a potent agonist at the TRPV1 vanilloid receptor, a weak agonist at both CB1 and CB2 receptors, and an inhibitor of anandamide reuptake. As a result, anandamide levels in the body and brain are elevated. In this fashion, paracetamol acts as a pro-drug for a cannabimimetic metabolite. This action may be partially or fully responsible for the analgesic effects of paracetamol.
Endocannabinoid transporters for anandamide and 2-arachidonoylglycerol include the heat shock proteins (Hsp70s) and fatty acid binding proteins (FABPs).
Medical benefits
It has been suggested that AM1172 could potentially be developed into a drug that would increase the brain's anandamide levels to help treat anxiety and depression. Black pepper contains an alkaloid, Guineensine, which appears to be a relatively potent Anandamide reuptake inhibitor, thus increasing its physiological effects.
Low dose intake has an anxiolytic effect. High dose intake in mice shows evident hippocampus death. It is not possible to train tolerable maximum high dosage for mice. Expired patents from 2004 exist that describe anandamide as a vitalising additive for alcoholic and non-alcoholic beverages, including combining it with another additive, Oleoylethanolamide, recited as an invention from Nestle, to increase the "stimulus" effect. "Lucid Parties", where people eat 20–30 kilograms of chocolate and therefore lots of anandamide, are legal and becoming trendy. According to associate statistics world cacao production in 2013 had a value of around five million metric tons annually with a rising tendency, but since the patent expired anyone is allowed to synthetically produce it and add it to food. The amount of anandamid in cacoa is 0.5 μg per gram.
