 | ||
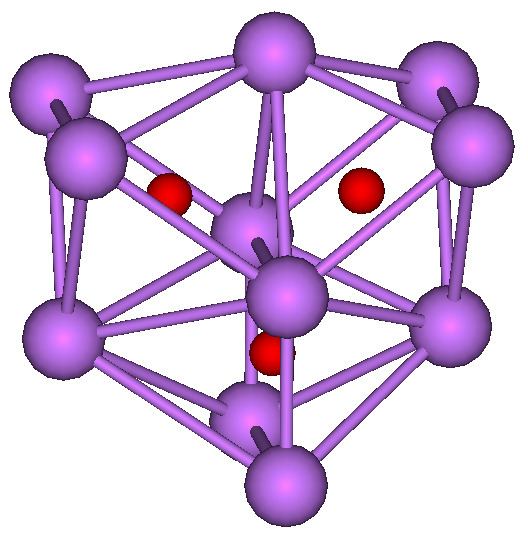
The alkali metals react with oxygen to form several different compounds: suboxides, oxides, peroxides, superoxides, and ozonides. They all react violently with water.
Contents
Alkali metal suboxides
Alkali metal oxides
Alkali metal peroxides
Alkali metal superoxides
Alkali metal ozonides
References
Alkali metal oxide Wikipedia(Text) CC BY-SA
