 | ||
Acid guanidinium thiocyanate-phenol-chloroform extraction (abbreviated AGPC) is a liquid–liquid extraction technique in biochemistry. It is widely used in molecular biology for isolating RNA (as well as DNA and protein in some cases). This method may take longer than a column-based system such as the silica-based purification, but has higher purity and the advantage of high recovery of RNA: an RNA column is typically unsuitable for purification of short (<200 nucleotides) RNA species, such as siRNA, miRNA, gRNA and tRNA.
Contents
It was originally devised by Piotr Chomczynski and Nicoletta Sacchi and published in 1987. The reagent is sold by Sigma-Aldrich by the name TRI Reagent; by Invitrogen under the name TRIzol; by Bioline as Trisure; and by Tel-Test as STAT-60.
How it works
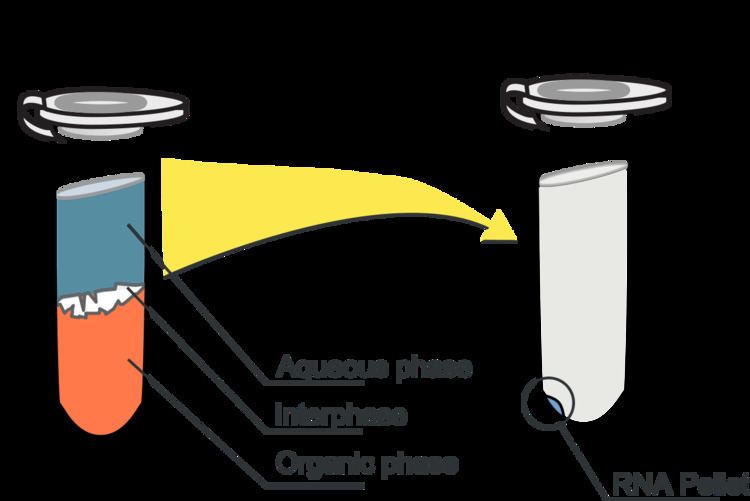
This method relies on phase separation by centrifugation of a mixture of the aqueous sample and a solution containing water-saturated phenol and chloroform, resulting in an upper aqueous phase and a lower organic phase (mainly phenol). Guanidinium thiocyanate, a chaotropic agent, is added to the organic phase to aid in the denaturation of proteins (such as those that strongly bind nucleic acids or those that degrade RNA). The nucleic acids (RNA and/or DNA) partition into the aqueous phase, while protein partitions into the organic phase. The pH of the mixture determines which nucleic acids get purified. Under acidic conditions (pH 4-6), DNA partitions into the organic phase while RNA remains in the aqueous phase. Under neutral conditions (pH 7-8), both DNA and RNA partition into the aqueous phase. In a last step, the nucleic acids are recovered from the aqueous phase by precipitation with 2-propanol or ethanol.
Guanidinium thiocyanate denatures proteins, including RNases, and separates rRNA from ribosomal proteins, while phenol, isopropanol and water are solvents with poor solubility. In the presence of chloroform or BCP (bromochloropropane), these solvents separate entirely into two phases that are recognized by their color: a clear, upper aqueous phase (containing the nucleic acids) and a lower phase (containing the proteins dissolved in phenol and the lipids dissolved in chloroform). Other denaturing chemicals such as 2-mercaptoethanol and sarcosine may also be used. The major downside is that phenol and chloroform are both hazardous and inconvenient materials, and the extraction is often laborious, so in recent years many companies now offer alternative ways to isolate DNA.
