ATC code None Synonyms LY2835219 PubChem CID 46220502 | Legal status Investigational CAS Number 1231929-97-7 ChemSpider 29340700 | |
 | ||
Fda breakthrough status for abemaciclib
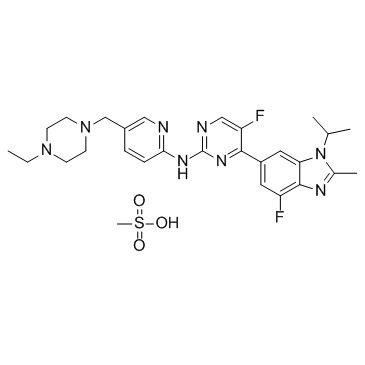
Abemaciclib (previously known as LY2835219) is a CDK inhibitor selective for CDK4 and CDK6. It is an investigational drug for various types of cancer developed by Eli Lilly. It was designated as a breakthrough therapy by the U.S. Food and Drug Administration in October 2015.
Contents
- Fda breakthrough status for abemaciclib
- Ongoing trial of abemaciclib and pembrolizumab in hr her2 negative breast cancer
- Clinical trials
- Chemistry
- References
Ongoing trial of abemaciclib and pembrolizumab in hr her2 negative breast cancer
Clinical trials
Successful Phase I and Phase II trials against breast cancer were announced in May and December 2014 respectively.
As of early 2016 Abemaciclib is involved in 3 Phase III clinical trials:
Chemistry
Abemaciclib may be synthesized in a 4 step manner using a Suzuki coupling, followed by a Buchwald–Hartwig amination with the final step being a reductive amination using the Leuckart reaction.
References
Abemaciclib Wikipedia(Text) CC BY-SA
