Formula C12H14N4C12H18Cl4N4(4HCl) | Molar mass 214.27 g/mol | |
 | ||
3,3'-Diaminobenzidine (DAB) is an organic compound that is both chemically and thermodynamically stable. This derivative of benzidine is a precursor to polybenzimidazole fiber. This fiber can be used to construct materials that are resistant to chemicals and high temperature. Fabrics made from these fibers do not melt when exposed to flame and do not burn. As its water-soluble tetrahydrochloride, DAB has been used in immunohistochemical staining of nucleic acids and proteins.
Contents

DAB is symmetric about the central carbon bond between both ring structures. In the crystal, the rings of each molecule are co-planar and the amine units connect molecules to form an intermolecular 3-dimensional hydrogen bond network.
Preparation
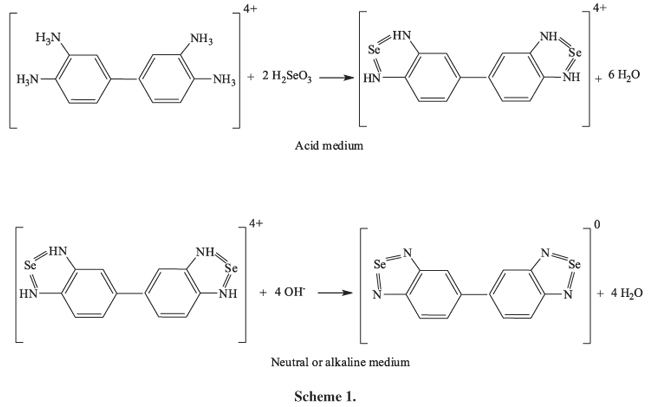
Diaminobenzidine, which is commercially available, is prepared by reacting 3,3'-dichlorobenzidine and ammonia with a copper catalyst at high temperature and pressure, followed by acidic workup.
An alternate synthesis route involves the diacylation of benzidine with acetic anhydride under basic conditions:
(NH2)C6H4C6H4(NH2) + 2(CH3CO)2O ⟶ (NHCOCH3)C6H4C6H4(NHCOCH3) + 2CH3CO2H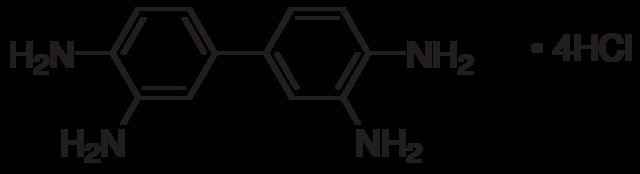
The diacetylated compound then undergoes nitration with nitric acid to produce an ortho-dinitro compound due to the ortho-directing acetyl substituents:
(NHCOCH3)C6H4C6H4(NHCOCH3) + 2HNO3 ⟶ (O2N)(NHCOCH3)C6H3C6H3(NHCOCH3)(NO2) + 2H2OThe acetyl groups are then removed through saponification:
(O2N)(NHCOCH3)C6H3C6H3(NHCOCH3)(NO2) + 2NaOH ⟶ (O2N)(NH2)C6H3C6H3(NH2)(NO2) + 2(NaOCOCH3)The dinitrobenzidine compound is then reduced with hydrochloric acid and iron to produce 3,3'-diaminobenzidine:
3(O2N)(NH2)C6H3C6H3(NH2)(NO2) + 12HCl + 10Fe0 ⟶ 3(NH2)2C6H3C6H3(NH2)2 + 4Fe2O3 + 6FeCl2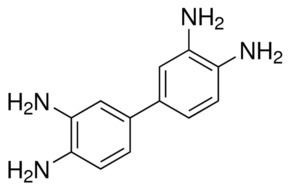
The reduction of the dinitrobenzidine compound can also proceed with tin(II) chloride instead of iron powder or with sodium dithionite in methanol.
Applications

Diaminobenzidine is oxidized by hydrogen peroxide in the presence of hemoglobin to give a dark-brown color. This is used to detect fingerprints in blood. The solubility of DAB in water allows for adaptability compared to other detection solutions which use toxic solvents. Improperly prepared tissue samples may give false positives In research, this reaction is used to stain cells that were prepared with hydrogen peroxidase enzyme, following common immunocytochemistry protocols. In research being done on Alzheimer's disease, Aβ protein amyloid plaques are targeted by a primary antibody, and subsequently by a secondary antibody, which is conjugated with a peroxidase enzyme. This will bind DAB as a substrate and oxidize it, producing an easily observable brown color. Plaques can then be quantified for further evaluation. One other method uses complexes of injected biocytin with avidin or streptavidin, biotin, and then peroxidase.
