Formula C6H4ClNO2 | Appearance Yellow crystals | |
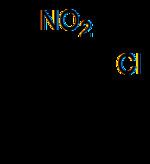 | ||
2-Nitrochlorobenzene is an organic compound with the formula ClC6H4NO2. It is a yellow crystalline solid that is important as a precursor to other compounds due to the two reactive sites present on the molecule.
Contents
Synthesis
Nitrochlorobenzene is typically synthesized by nitration of chlorobenzene in the presence of sulfuric acid:
C6H5Cl + HNO3 → O2NC6H4Cl + H2OThis reaction affords a mixture of isomers. Using an acid ratio of 30/56/14, the product mix is typically 34-36% 2-nitrochlorobenzene and 63-65% 4-nitrochlorobenzene, with only about 1% 3-nitrochlorobenzene.
Reactions
Alkylation and electrophilic aromatic substitution can occur at the chlorinated carbon center, and a diverse array of reactions can be carried out using the nitro group. 2-Nitrochlorobenzene can be reduced to 2-chloroaniline with Fe/HCl mixture, the Bechamp reduction.
Applications
2-Nitrochlorobenzene is not valuable in itself but is a precursor to other useful compounds. The compound is particularly useful because both of its reactive sites can be utilized to create further compounds that are mutually ortho. Its derivative 2-chloroaniline is a precursor to 3,3’-dichlorobenzidine, itself a precursor to many dyes and pesticides.
