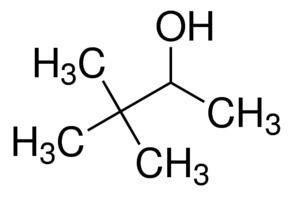
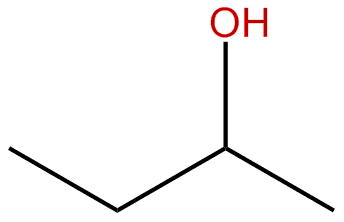
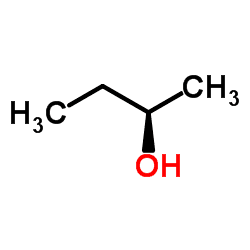
Related compounds Density 806 kg/m³ | Formula C4H10O Molar mass 74.122 g/mol | |
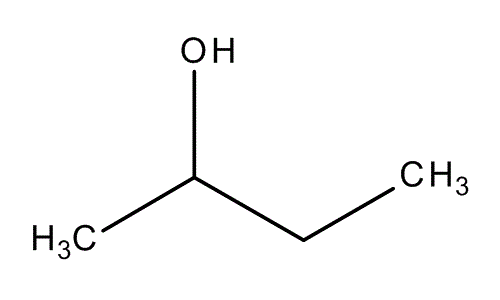 | ||
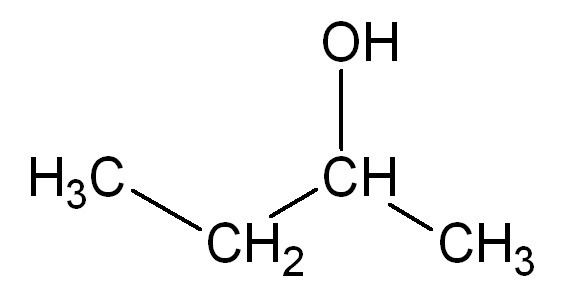
2-Butanol, or sec-butanol, is an organic compound with formula CH3CH(OH)CH2CH3. This secondary alcohol is a flammable, colorless liquid that is soluble in 3 parts water and completely miscible with polar organic solvents such as ethers and other alcohols. It is produced on a large scale, primarily as a precursor to the industrial solvent methyl ethyl ketone. 2-Butanol is chiral and thus can be obtained as either of two stereoisomers designated as (R)-(−)-2-butanol and (S)-(+)-2-butanol. It is normally found as an equal mixture of the two stereoisomers — a racemic mixture.
Contents
Manufacture and applications
2-Butanol is manufactured industrially by the hydration of 1-butene or 2-butene:
Sulfuric acid is used as a catalyst for this conversion.

In the lab it can be prepared via Grignard reaction by reacting ethylmagnesium bromide with acetaldehyde in dried diethyl ether or tetrahydrofuran.
Although some 2-butanol is used as a solvent, it is mainly converted to butanone ("MEK"), which is an important industrial solvent and found in many domestic cleaning agents and paint removers. Volatile esters of 2-butanol have pleasant aromas and are used in small amounts as perfumes or in artificial flavors.
Solubility
The solubility of 2-butanol is incorrect in most sources, including some of the most well-known references such as the Merck Index, the CRC Handbook of Chemistry and Physics, and Lange's Handbook of Chemistry. Even the International Programme on Chemical Safety lists the wrong solubility. This widespread error originated because of Beilstein's Handbuch der Organischen Chemie (Handbook of Organic Chemistry). This work cites a false solubility of 12.5 g/100 ml water. Many other sources used this solubility, which has snowballed into a widespread error in the industrial world. The correct data (35.0 g/100 ml at 20 °C, 29 g/100 ml at 25 °C, and 22 g/100 ml at 30 °C) was first published in 1886 by Alexejew and then similar data was reported by other scientists including Dolgolenko and Dryer in 1907 and 1913, respectively.
Safety
Several explosions have been reported during the conventional distillation of 2-butanol, apparently due to the buildup of peroxides with the boiling point higher than that of pure alcohol (and therefore concentrating in the still pot during distillation). As alcohols, unlike ethers, are not widely known to be capable of forming peroxide impurities, the danger is likely to be overlooked.