3DMet B00214 Formula C3H10N2 Molar mass 74.1249 g/mol | Abbreviations TMEDA Density 880 kg/m³ Appearance Colourless liquid | |
 | ||
Related alkanamines Related compounds | ||
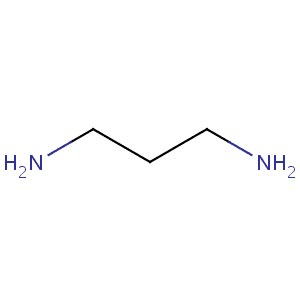
1,3-Diaminopropane, also known as trimethylenediamine, is a simple diamine with the formula (CH2)3(NH2)2. A colourless liquid with a fishy odor, it is soluble in water and many polar organic solvents. It is isomeric with 1,2-diaminopropane. Both are building blocks in the synthesis of heterocycles, such as those used in textile finishing, and coordination complexes. It is prepared by the amination of acrylonitrile followed by hydrogenation of the resulting aminopropionitrile.
The potassium salt was used in the alkyne zipper reaction.
Known uses of 1,3-Diaminopropane is in the synthesis of piroxantrone and Losoxantrone.
Safety
1,3-Diaminopropane is toxic on skin exposure with an LD50 of 177 mg kg−1 (dermal, rabbit)
References
1,3-Diaminopropane Wikipedia(Text) CC BY-SA
