ATC code None Synonyms MK-8931 PubChem CID 51352361 ChemSpider ID 31399364 | Legal status Investigational CAS Number 1286770-55-5 UNII J1I0P6WT7T | |
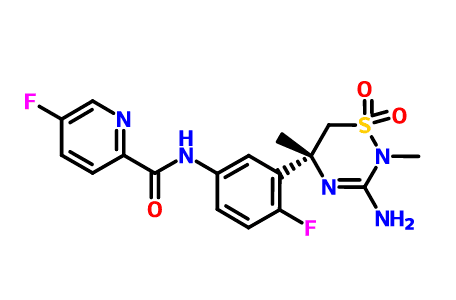 | ||
Verubecestat
Verubecestat (MK-8931) is an experimental drug for the treatment of Alzheimer's disease. It is an inhibitor of beta-secretase 1 (BACE1).
In April 2012 phase I clinical results were announced. Phase 1b results have also been reported.
As of December 2016 it is in two phase 2/3 clinical trials that have progressed to phase 3. EPOCH, was to complete data collection for the primary outcome measure by June 2017.
In February 2017, Merck halted its late-stage trial of verubecestat for mild to moderate Alzheimer's disease after it was reported as having "virtually no chance of finding a positive clinical effect" according to an independent panel of experts. The results of Merck's trial of verubecestat on patients with prodromal (early stage) Alzheimer's are still expected in February 2019.
