Formula VO Melting point 1,789 °C Boiling point 2,627 °C | Molar mass 66.9409 g/mol Density 5.76 g/cm³ Pubchem 24411 | |
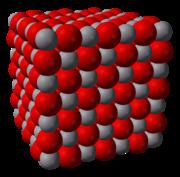 | ||
Appearance grey solid with metallic lustre | ||
Vanadium(II) oxide, VO, is one of the many oxides of vanadium. VO is a long-lived, electronically neutral reagent chemical. It adopts a distorted NaCl structure and contains weak V-V metal to metal bonds. As shown by band theory, VO is a conductor of electricity due to its partially filled conduction band and delocalisation of electrons in the t2g orbitals. VO is a non-stoichiometric compound, its composition varying from VO0.8 to VO1.3.
References
Vanadium(II) oxide Wikipedia(Text) CC BY-SA
