Examples W(CH3)6 Coordination number 6 | Steric number 6 Bond angle(s) 95°, 85° | |
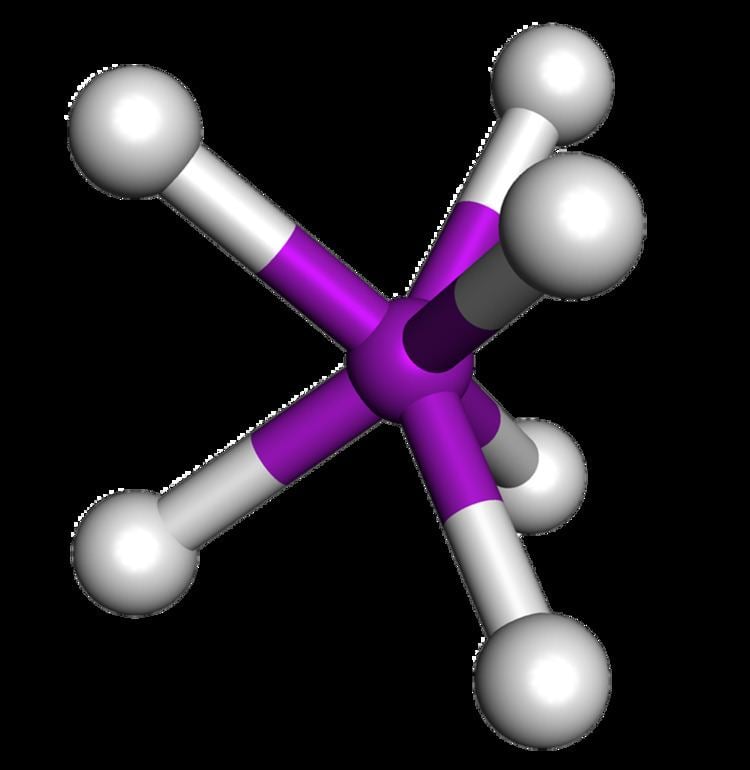 | ||
In chemistry, the trigonal prismatic molecular geometry describes the shape of compounds where six atoms, groups of atoms, or ligands are arranged around a central atom, defining the vertices of a triangular prism.
Examples
Hexamethyltungsten (W(CH3)6) was the first example of a molecular trigonal prismatic complex.
Some other transition metals have trigonal prismatic hexamethyl complexes, including both neutral molecules such as Mo(CH3)6 and Re(CH3)6 and ions such as Ta(CH
3)−
6 and Zr(CH
3)2−
6.
The complex Mo(S−CH=CH−S)3 is also trigonal prismatic, with each S−CH=CH−S group acting as a bidentate ligand with two sulfur atoms binding the metal atom. Here the coordination geometry of the six sulfurs around the molybdenum is similar to that in the extended structure of molybdenum disulfide (MoS2).
