Related compounds Molar mass 1,244.24 g/mol | Formula C72H60P4Pt Appearance yellow crystals | |
 | ||
Tetrakis(triphenylphosphine)platinum(0) is the chemical compound with the formula Pt(P(C6H5)3)4, often abbreviated Pt(PPh3)4. The bright yellow compound is used as a precursor to other platinum complexes.
Contents
Structure and behavior
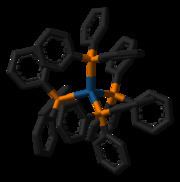
The molecule is tetrahedral, with point group symmetry of Td, as expected for a four-coordinate metal complex of a metal with the d10 configuration. Even though this complex follows the 18 electron rule, it dissociates triphenylphosphine in solution to give the 16e− derivative containing only three PPh3 ligands:
Pt(PPh3)4 → Pt(PPh3)3 + PPh3Synthesis and reactions
The complex is typically prepared in one-pot reaction from potassium tetrachloroplatinate(II). Reduction of this platinum(II) species with alkaline ethanol in the presence of excess triphenylphosphine affords the product as a precipitate. The reaction occurs in two distinct steps. In the first step, PtCl2(PPh3)2 is generated. In the second step, this platinum(II) complex is reduced. The overall synthesis can be summarized as:
K2[PtCl4] + 2KOH + 4PPh3 + C2H5OH → Pt(PPh3)4 + 4KCl + CH3CHO + 2H2OPt(PPh3)4 reacts with oxidants to give platinum(II) derivatives:
Pt(PPh3)4 + Cl2 → cis-PtCl2(PPh3)2 + 2 PPh3Mineral acids give the corresponding hydride complexes:
Pt(PPh3)4 + HCl → trans-PtCl(H)(PPh3)2 + 2 PPh3The reaction with oxygen affords a dioxygen complex:
Pt(PPh3)4 + O2 → Pt(η2-O2)(PPh3)2 + 2 PPh3This complex is a precursor to the ethylene complex
Pt(η2-O2)(PPh3)2 + C2H4 → Pt(η2-C2H4)(PPh3)2 + "NaBH2(OH)2"