Formula Cl8K4Mo2 Density 2.54 g/cm³ | Molar mass 631.9 g/mol Appearance red crystals | |
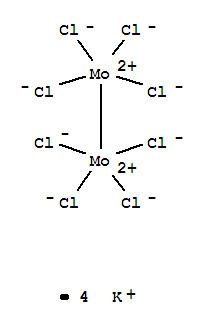 | ||
Potassium octachlorodimolybdate (systematically named potassium bis(tetrachloridomolybdate)(Mo–Mo)(4−)) is an inorganic compound with the chemical formula K
4Mo
2Cl
8 (also written as K
4[Cl
4MoMoCl
4]). It is known as a red-coloured, microcrystalline solid. The anion is of historic interest as one of the earliest illustrations of a quadruple bonding. The salt is usually obtained as the pink-coloured dihydrate.
The compound is prepared in two steps from molybdenum hexacarbonyl:
2 Mo(CO)6 + 4 HO2CCH3 → Mo2(O2CCH3)4 + 2 H2 + 12 COMo2(O2CCH3)4 + 4 HCl + 4 KCl → K4Mo2Cl8 + 4 HO2CCH3The reaction of the acetate with HCl was first described as providing trimolybdenum compounds, but subsequent crystallographic analysis confirmed that the product contains the Mo2Cl4–
8 ion with D4h symmetry. The Mo–Mo distance is 2.14 Å.
References
Potassium octachlorodimolybdate Wikipedia(Text) CC BY-SA
