Abbreviations PMHS Density 1.06 g/cm³ | Formula (CH3(H)SiO)n | |
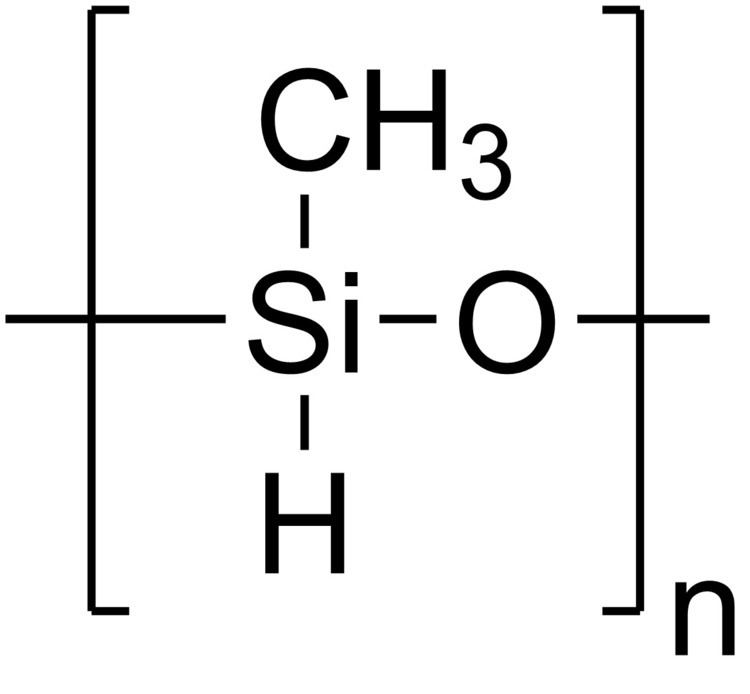 | ||
Polymethylhydrosiloxane (PMHS) is a polymer with the general structure -(CH3(H)Si-O)-. It is used in organic chemistry as a mild and stable reducing agent easily transferring hydrides to metal centers. A variety of related materials are available under the following CAS registry numbers 9004-73-3, 16066-09-4, 63148-57-2, 178873-19-3. These include the tetramer ((MeSiHO)4), copolymers of dimethysiloxane and methylhydrosiloxane, and trimethylsilyl terminated materials.
This material is prepared by the hydrolysis of methyldichlorosilane:
n MeSiHCl2 + n H2O → (MeSiHO)n + 2n HClThe related polymer polydimethylsiloxane (PDMS) is made similarly, but lacking Si-H bonds, it exhibits no reducing properties.
Illustrative of its use, PMHS is used for in situ conversion of tributyltin oxide to tributyltin hydride:
2"(MeSiH)" + (Bu3Sn)2O → "(Me2Si2O" + 2 Bu3SnH