Formula LiCoO2 | Molar mass 97.87 g/mol | |
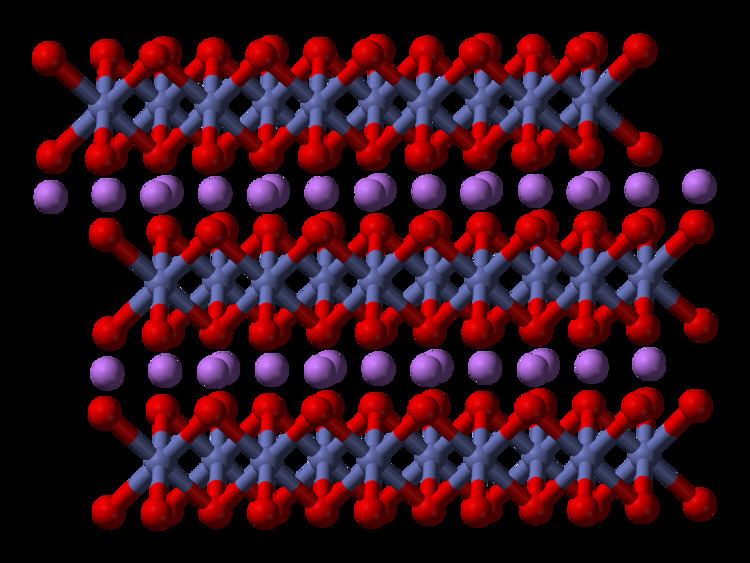 | ||
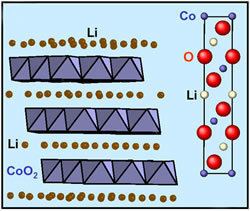
Lithium cobalt oxide (LiCoO
2) is a chemical compound commonly used in the positive electrodes of lithium-ion batteries. The structure of LiCoO
2 has been studied with numerous techniques including x-ray diffraction, electron microscopy, neutron powder diffraction, and EXAFS: it consists of layers of lithium that lie between slabs of octahedra formed by cobalt and oxygen atoms. The space group is
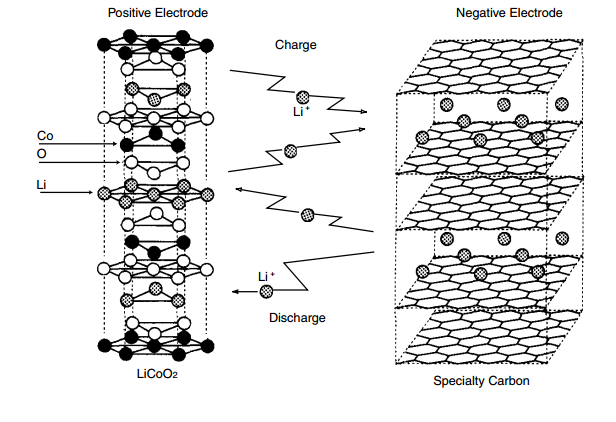
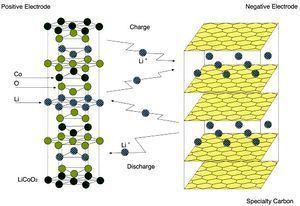
Batteries produced with LiCoO
2 cathodes have very stable capacities, but have lower capacities and power than cathodes based on nickel-cobalt-aluminum (NCA) oxides. Issues with thermal stability are better for LiCoO
2 cathodes than other nickel-rich chemistries although not significantly. This makes LiCoO
2 batteries susceptible to thermal runaway in cases of abuse such as high temperature operation (>130 °C) or overcharging. At elevated temperatures, LiCoO
2 decomposition generates oxygen, which then reacts with the organic electrolyte of the cell. This is a safety concern due to the magnitude of this highly exothermic reaction, which can spread to adjacent cells or ignite nearby combustible material. In general, this is seen for many lithium ion battery cathodes.
The compound's usefulness as an intercalation electrode was discovered in 1980 by John B. Goodenough's research group at Oxford.