 | ||
The Japp–Klingemann reaction is a chemical reaction used to synthesize hydrazones from β-keto-acids (or β-keto-esters) and aryl diazonium salts. The Reaction is named after the chemists Francis Robert Japp and Felix Klingemann.
The hydrazone products of the Japp–Klingemann reaction are most often used as intermediates in syntheses of more complex organic molecules. For example, a phenylhydrazone product can be heated in the presence of strong acid to produce an indole via the Fischer indole synthesis.
Reaction mechanism
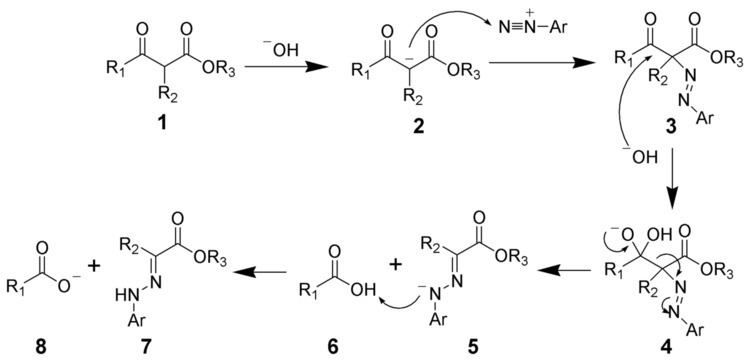
To illustrate the mechanism, the Japp-Klingemann ester variation will be considered. The first step is the deprotonation of the β-keto-ester. The nucleophilic addition of the enolate anion 2 to the diazonium salt produces the azo compound 3. Intermediate 3 has been isolated in rare cases. However, in most cases, the hydrolysis of intermediate 3 produces a tetrahedral intermediate 4, which quickly decomposes to release the carboxylic acid 6. After hydrogen exchange, the final hydrazone 7 is produced.
