Formula C26H16 | Molar mass 328.41 g/mol | |
 | ||
Hexacene is an aromatic molecule consisting of six linearly-fused benzene rings. Hexacene and its derivatives are investigated for potential applications as organic semiconductor.
Hexacene is one of a series of linear acenes, the previous one being pentacene (five fused benzene rings) and the next one being heptacene (seven fused benzene rings).
Hexacene is very unstable, being highly reactive in the 6, 15 positions due to pi electron localization. Its spectra must be collected on freshly purified samples handled in an inert atmosphere such as nitrogen or argon. Only with very large substituents it is possible to isolate an hexacene for instance the compound 6,15-bis(tri-t-butylsilylethynyl)hexacene which melts with decomposition at 96 °C.
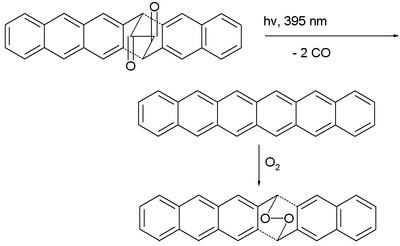
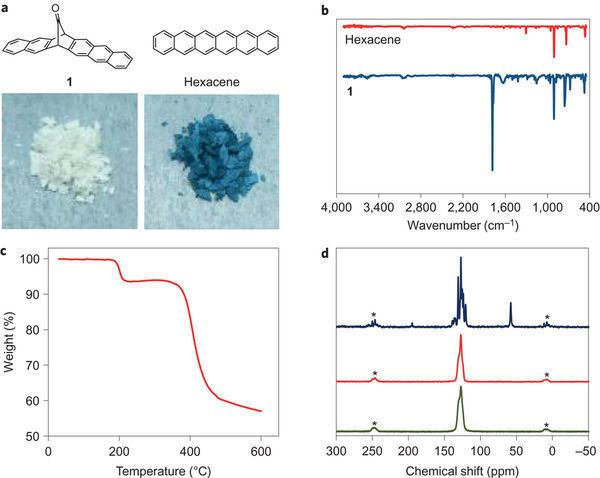
The first reported synthesis of hexacene itself dates back to 1939. In 1955 the compound is synthesized by dehydrogenation of hexacosadehydrohexacene by palladium on carbon. In 1982 hexacene is reported as a bluish green compound with decomposition at 380 degrees. In 2007, a research group claimed the first reproducible unsubstituted hexacene synthesis (invalidating the previous claims) based on photochemical decarbonylation of a diketone precursor:

The compound could not be isolated: it dimerized even at concentration as low as 10−4 M and reacted in solution with oxygen to the organic peroxide. In a PMMA polymer matrix on the other hand these side-reactions were limited and the compound survived up to 12 hours. The next homologue heptacene also studied by this group, was even more unstable (decomposes within 4 hours).
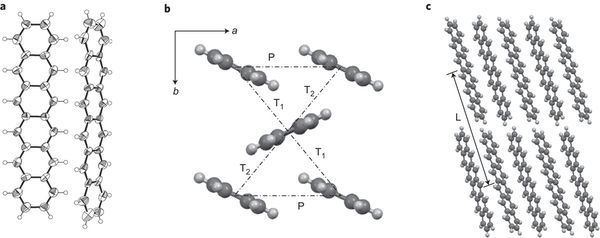
The synthesis of hexacene has also been reported from a monoketone precursor. by solid state synthesis at 180 °C as a crystalline solid. This solid was reported stable in the dark for up to a month, attributed to its herringbone packing preventing dimerization.
