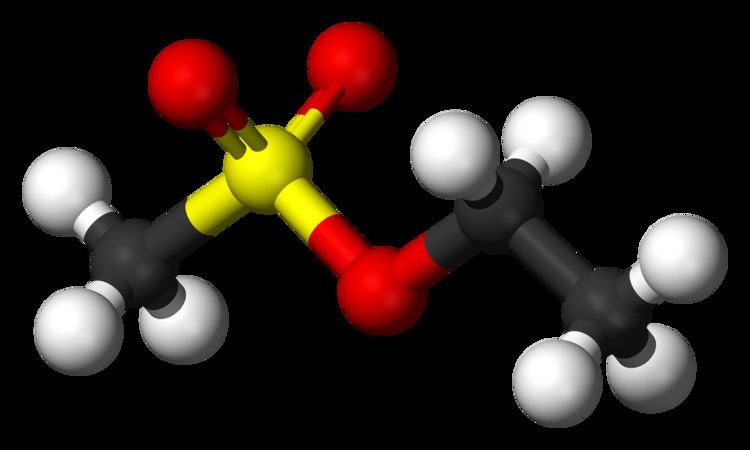
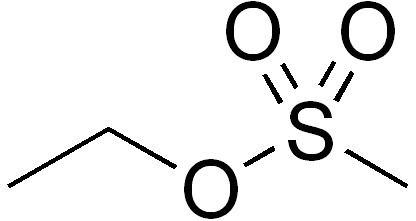
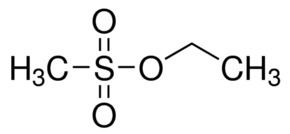
Abbreviations EMS Molar mass 124.16 g/mol Appearance Clear colorless liquid | Formula CH3SO3C2H5 Density 1.15 g/cm³ | |
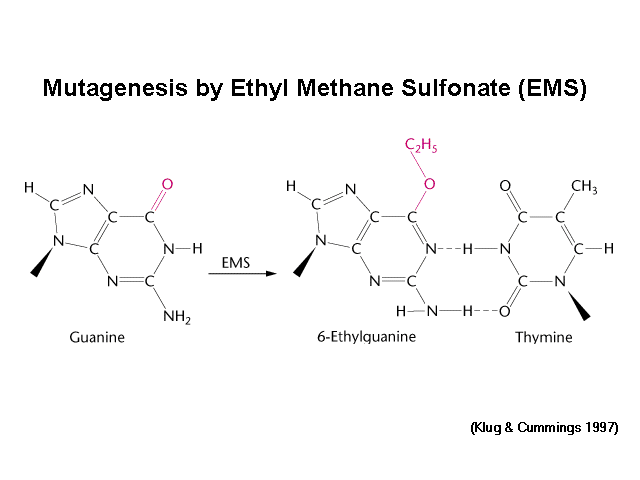 | ||
Ethyl methanesulfonate (EMS) is a mutagenic, teratogenic, and possibly carcinogenic organic compound with formula C3H8SO3. It produces random mutations in genetic material by nucleotide substitution; particularly by guanine alkylation. This typically produces only point mutations. It can induce mutations at a rate of 5x10−4 to 5x10−2 per gene without substantial killing. The ethyl group of EMS reacts with guanine in DNA, forming the abnormal base O6-ethylguanine. During DNA replication, DNA polymerases that catalyze the process frequently place thymine, instead of cytosine, opposite O6-ethylguanine. Following subsequent rounds of replication, the original G:C base pair can become an A:T pair (a transition mutation). This changes the genetic information, is often harmful to cells, and can result in disease.
EMS is often used in genetics as a mutagen. Mutations induced by EMS can then be studied in genetic screens or other assays.