 | ||
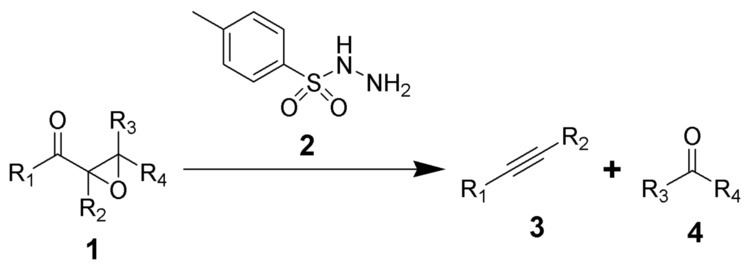
The Eschenmoser fragmentation, first published in 1967, is the chemical reaction of α,β-epoxyketones (1) with aryl sulfonylhydrazines (2) to give alkynes (3) and carbonyl compounds (4). This reaction is named after the Swiss chemist Albert Eschenmoser, who devised it in collaboration with an industrial research group around Günther Ohloff in Firmenich for the production of muscone and related macrocyclic musks. Hence, the reaction is also known as Eschenmoser–Ohloff fragmentation. In the same year, i.e. 1967, Masato Tanabe et al. of SRI International published an article detailing the reaction, and thus, some also refer to it as the Eschenmoser–Tanabe fragmentation.
Several examples exist in the literature, and the reaction is also carried out on industrial scale.
Reaction mechanism
The mechanism of the Eschenmoser fragmentation begins with the condensation of an α,β-epoxyketone (1) with an aryl sulfonylhydrazine (2) to afford the intermediate hydrazone (3). This hydrazone can either be protonated at the expoxide oxgygen or deprotonated at the sulfonamide nitrogen to initiate the fragmentation, and thus the fragmentation is catalyzed by acids or bases. Most common reaction conditions, however, are treatment with acetic acid in dichloromethane. The proton transfer leads to intermediate 4, which undergoes the key fragmentation to alkyne (6) and the corresponding carbonyl compound (7). Driving force is the formation of molecular nitrogen.
Besides this standard course, there is also a radical variant of this α,β-enone→alkynone fragmentation, which employs 1,3-dibromo-5,5-dimethylhydantoin (DDH) in sec-butanol. Therefore, an epoxide is not required. The α,β-unsaturated hydrazone is brominated by DDH in allylic position at the sulfonamide nitrogen, which constitutes the capto-dative stabilized radical position, and the bromide ion becomes the leaving group in the following nucleophilic attack of an alcoholate ion. This Fehr–Ohloff–Büchi variant of the Eschenmoser–Ohloff fragramentation thus avoids the epoxidations step, which in case of sterically demanding substrates often leads to low yields of the classical Eschenmoser fragmentation.
