Appearance red solid | ||
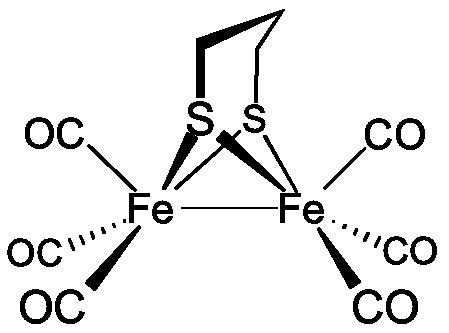 | ||
Diiron propanedithiolate hexacarbonyl is the organoiron complex with the formula Fe2(S2C3H6)(CO)6. It is a red diamagnetic solid. It adopts a symmetrical structure with six terminal CO ligands. The complex is a precursor to hydrogenase mimics.
It is prepared by the reaction of 1,3-propanedithiol with triiron dodecarbonyl:
2 Fe3(CO)12 + 3 C3H6(SH)2 → 3 Fe2(S2C3H6)(CO)6 + 3 H2 + 6 COIn general, the CO ligands can be substituted by cyanide, phosphines, isocyanides, N-heterocyclic carbenes, and other donor ligands. Monosubstitution can be achieved through an in situ generation of the acetonitrile complex.
Upon irradiation of Fe2(S2C3H6)(CO)6 with ultraviolet (UV) light, CO-photolysis occurs with the transient formation of the unsaturated species followed by the formation of the solvent adduct.
References
Diiron propanedithiolate hexacarbonyl Wikipedia(Text) CC BY-SA
