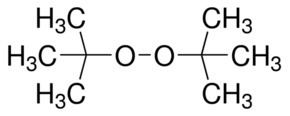
Formula C8H18O2 Density 800 kg/m³ | Molar mass 146.23 g/mol | |
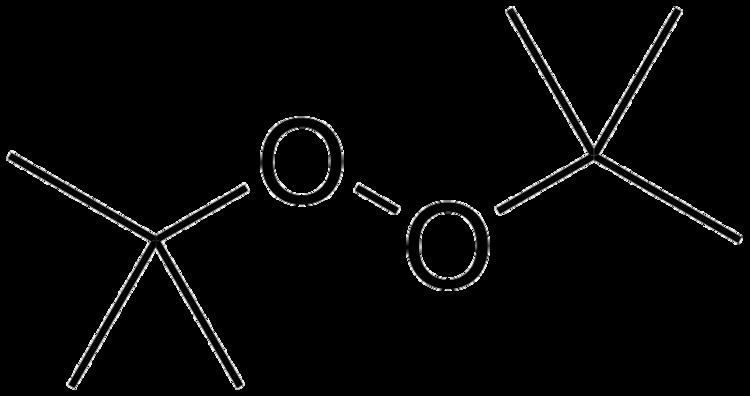 | ||
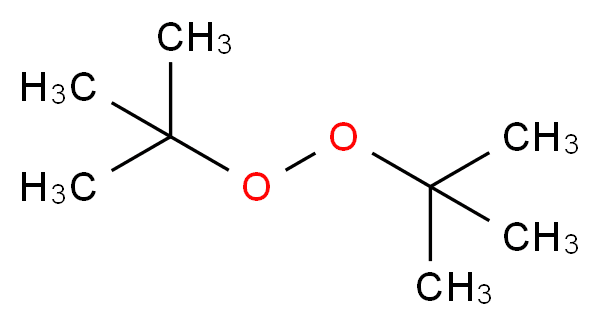
Di-tert-butyl peroxide or DTBP is an organic compound consisting of a peroxide group bonded to two tert-butyl groups. It is one of the most stable organic peroxides. It is a colorless liquid.
Reactions
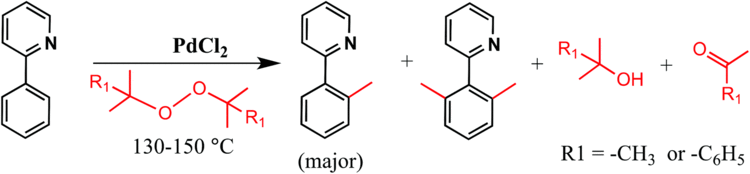
The peroxide bond undergoes homolysis at temperatures >100 °C. For this reason di-tert-butyl peroxide is commonly used as a radical initiator in organic synthesis and polymer chemistry. The decomposition reaction proceeds via the generation of methyl radicals.
(CH3)3COOC(CH3)3 → 2 (CH3)3CO.(CH3)3CO. → (CH3)2CO + CH3.2 CH3. → CH3-CH3
DTBP can in principle be used in engines where oxygen is limited, since the molecule supplies both the oxidizer and the fuel.
References
Di-tert-butyl peroxide Wikipedia(Text) CC BY-SA
