Symbol Citrate_synt InterPro IPR002020 SCOP 1csc | Pfam PF00285 PROSITE PDOC00422 SUPERFAMILY 1csc | |
 | ||
In molecular biology, the citrate synthase family of proteins includes the enzymes citrate synthase EC 2.3.3.1, and the related enzymes 2-methylcitrate synthase EC 2.3.3.5 and ATP citrate synthase EC 2.3.3.8.
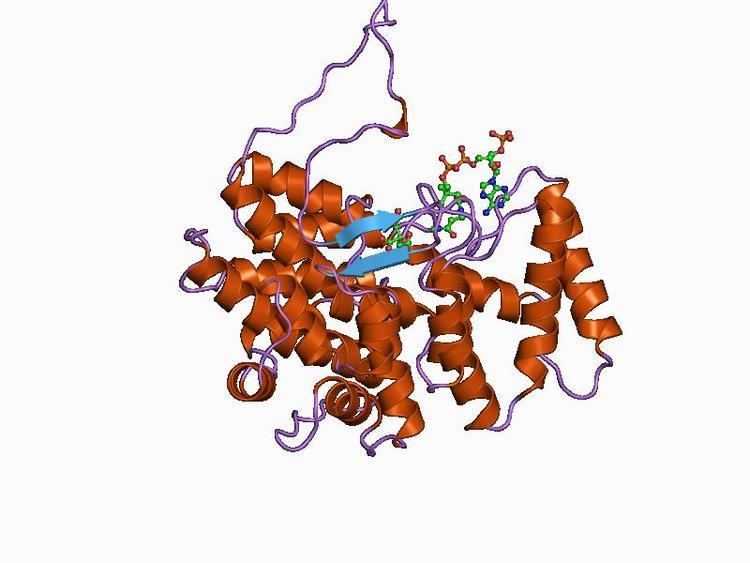
Citrate synthase is a member of a small family of enzymes that can directly form a carbon-carbon bond without the presence of metal ion cofactors. It catalyses the first reaction in the Krebs' cycle, namely the conversion of oxaloacetate and acetyl-coenzyme A into citrate and coenzyme A. This reaction is important for energy generation and for carbon assimilation. The reaction proceeds via a non-covalently bound citryl-coenzyme A intermediate in a 2-step process (aldol-Claisen condensation followed by the hydrolysis of citryl-CoA).
Citrate synthase enzymes are found in two distinct structural types: type I enzymes (found in eukaryotes, Gram-positive bacteria and archaea) form homodimers and have shorter sequences than type II enzymes, which are found in Gram-negative bacteria and are hexameric in structure. In both types, the monomer is composed of two domains: a large alpha-helical domain consisting of two structural repeats, where the second repeat is interrupted by a small alpha-helical domain. The cleft between these domains forms the active site, where both citrate and acetyl-coenzyme A bind. The enzyme undergoes a conformational change upon binding of the oxaloacetate ligand, whereby the active site cleft closes over in order to form the acetyl-CoA binding site. The energy required for domain closure comes from the interaction of the enzyme with the substrate. Type II enzymes possess an extra N-terminal beta-sheet domain, and some type II enzymes are allosterically inhibited by NADH.
2-methylcitrate synthase catalyses the conversion of oxaloacetate and propanoyl-CoA into (2R,3S)-2-hydroxybutane-1,2,3-tricarboxylate and coenzyme A. This enzyme is induced during bacterial growth on propionate, while type II hexameric citrate synthase is constitutive.
ATP citrate synthase (also known as ATP citrate lyase) catalyses the MgATP-dependent, CoA-dependent cleavage of citrate into oxaloacetate and acetyl-CoA, a key step in the reductive tricarboxylic acid pathway of CO2 assimilation used by a variety of autotrophic bacteria and archaea to fix carbon dioxide. ATP citrate synthase is composed of two distinct subunits. In eukaryotes, ATP citrate synthase is a homotetramer of a single large polypeptide, and is used to produce cytosolic acetyl-CoA from mitochondrial produced citrate.
