 | ||
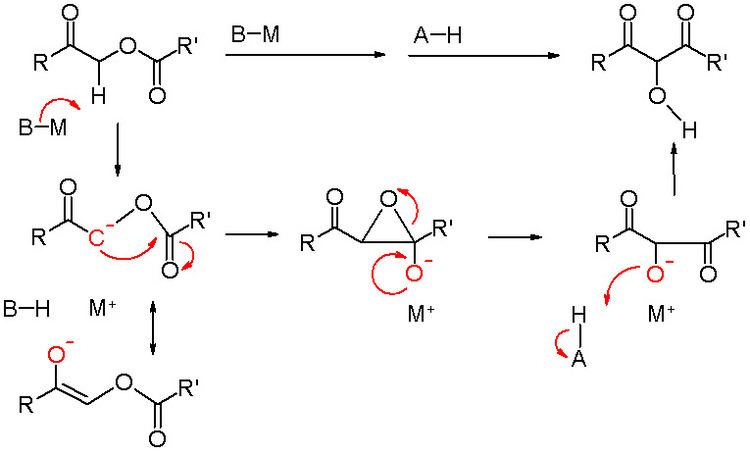
The Chan rearrangement is a chemical reaction that involves rearranging an acyloxy acetate (1) in the presence of a strong base to a 2-hydroxy-3-keto-ester (2).[1]
This procedure was rediscovered and employed in the Holton Taxol total synthesis.[2]
Reaction mechanism
The methylene bridge in the reactant with adjacent carbonyl and acetyl substituents is acidic and can be deprotonated by strong non-nucleophilic bases such as lithium tetramethylpiperidide or lithium diisopropylamide (LDA) as in an aldol reaction. The thus formed enolate then gives a nucleophilic acyl substitution with the adjacent carbonyl of the acetyl group through a short lived intermediate oxirane. Acidic workup liberates the free hydroxyl group.
References
Chan rearrangement Wikipedia(Text) CC BY-SA
