Molar mass 211.26 g/mol Melting point 220 °C | Formula C14H13NO Appearance Red crystals | |
 | ||
Brooker's merocyanine partitioned between water and octanol
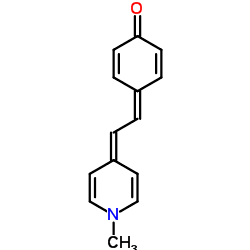
Brooker's merocyanine (1-methyl-4-[(oxocyclohexadienylidene)ethylidene]-1,4-dihydropyridine, MOED) is an organic dye belonging to the class of merocyanines.
Contents
- Brookers merocyanine partitioned between water and octanol
- Solvatochromic effects
- Uses
- Synthesis
- References
MOED is notable for its solvatochromic properties, meaning it changes color depending on the solvent in which it is dissolved.
As shown in the structural formula, MOED can exist in two resonance forms: A neutral molecule and a zwitterion. Research indicates that the zwitterion form is most representative when the compound exists in polar solvents such as water, and the neutral form when it exists in nonpolar solvents such as chloroform.
Solvatochromic effects

When MOED is dissolved in various liquids, its colour will vary, depending on the solvent and its polarity. In general, the more polar the solvent, the shorter the wavelengths of the light absorbed will be. When light of a certain colour (wavelength) is absorbed, the solution will appear in the complementary colour of the one absorbed. Therefore, in water, a highly polar solvent, MOED appears yellow (corresponding to absorbed blue light of wavelengths 435-480 nm), but is purple or blue (corresponding to absorbed green to yellow light of wavelengths 560-595 nm) in acetone, a less polar solvent. The effect stems in part from the stabilization of the ground state of the merocyanine molecule in polar solvents, which increases the energy gap between the ground state and excited states, which corresponds to shorter wavelengths (increased energy) of the absorbed light.
Uses
Because of its solvatochromic properties MOED, and solvatochromic dyes in general, are useful as solvent polarity indicators, and for creating solutions that absorb light at a specific frequency. Additional potential areas of use include pH sensors and transition metal cation indicators. Research into merocyanine dyes is ongoing.
Synthesis
Brooker's merocyanine can be prepared beginning with the methylation of 4-methylpyridine to produce 1,4-dimethylpyridinium iodide. Base catalyzed reaction with 4-hydroxybenzaldehyde and subsequent intramolecular dehydration provides Brooker's merocyanine.
