 | ||
Bredt's rule is an empirical observation in organic chemistry that states that a double bond cannot be placed at the bridgehead of a bridged ring system, unless the rings are large enough. The rule is named after Julius Bredt.
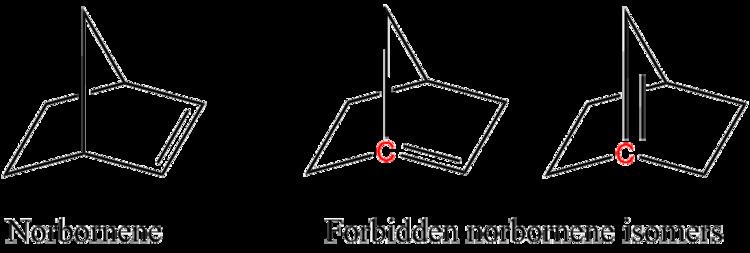
For example, two of the following isomers of norbornene violate Bredt's rule, which makes them too unstable to prepare:
In the figure, the bridgehead atoms involved in Bredt's rule violation are highlighted in red.
Bredt's rule is a consequence of the fact that having a double bond on a bridgehead would be equivalent to having a trans double bond on a ring, which is not stable for small rings (fewer than eight atoms) due to a combination of ring strain, and angle strain (nonplanar alkene). The p orbitals of the bridgehead atom and adjacent atoms are orthogonal and thus are not aligned properly for the formation of pi bonds.
Bredt's rule can be useful for predicting which isomer is obtained from an elimination reaction in a bridged ring system. It can also be applied to reaction mechanisms that go via carbocations and, to a lesser degree, via free radicals, because these intermediates, like carbon atoms involved in a double bond, prefer to have a planar geometry with 120 degree angles and sp2 hybridization.
An anti-bredt molecule is one that is found to exist and be stable (within certain parameters) despite this rule. A recent (2006) example of such a molecule is 2-quinuclidonium tetrafluoroborate. Bridgehead double bonds can be found in some natural products; see the article by Mak, Pouwer and Williams for a comprehensive review of natural products with anti-Bredt and bridgehead double bonds.
History
The first publication of what would later become known as Bredt's rule was in an article by Julius Bredt in 1924 about the chemistry of naturally occurring bicyclic terpenes. For an extensive review of this topic, see the article by Shea.
