 | ||
Antarafacial and suprafacial are two topological concepts in organic chemistry describing the relationship between two simultaneous chemical bond making and/or bond breaking processes in or around a reaction center. The reaction center can be a p-orbital, a conjugated system or even a sigma bond.
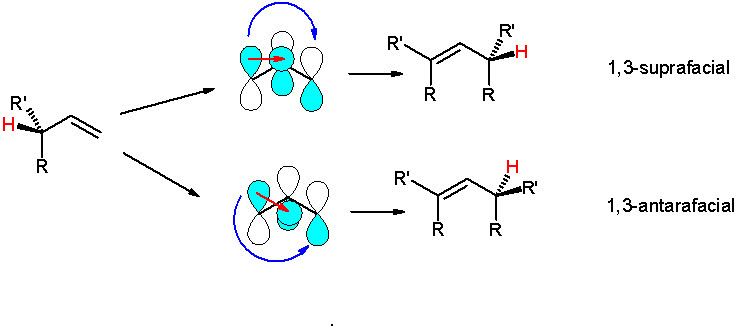
Many sigmatropic reactions and cycloadditions can be either suprafacial or antarafacial, and this determines the stereochemistry.
An example is the [1,3]-hydride shift, in which the interacting frontier orbitals are the allyl free radical and the hydrogen 1s orbitals. The suprafacial shift is symmetry-forbidden because orbitals with opposite algebraic signs overlap. The symmetry allowed antarafacial shift would require a strained transition state and is also unlikely. In contrast a symmetry allowed and suprafacial [1,5]-hydride shift is a common event.
