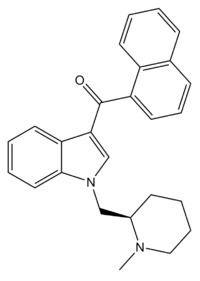
CAS Number 137642-54-7 ChemSpider 26231035 3D model (Jmol) Interactive image | PubChem CID 9929889 Formula C26H26N2O Molar mass 382.497 g/mol | |
 | ||
Legal status AU: S9 (Prohibited)CA: Schedule IIDE: Anlage II (Prohibited)NZ: Temporary Class | ||
AM-1220 is a drug that acts as a potent and moderately selective agonist for the cannabinoid receptor CB1, with around 19x selectivity for CB1 over the related CB2 receptor. It was originally invented in the early 1990s by a team led by Thomas D'Ambra at Sterling Winthrop, but has subsequently been researched by many others, most notably the team led by Alexandros Makriyannis at the University of Connecticut. The (piperidin-2-yl)methyl side chain of AM-1220 contains a stereocenter, so there are two enantiomers with quite different potency, the (R) enantiomer having a Ki of 0.27nM at CB1 while the (S) enantiomer has a much weaker Ki of 217nM. A number of related compounds are known with similar potent cannabinoid activity, with modifications such as substitution of the indole ring at the 2- or 6- positions, the naphthoyl ring substituted at the 4- position or replaced by substituted benzoyl rings or other groups, or the 1-(N-methylpiperidin-2-ylmethyl) group replaced by similar heterocyclic groups such as N-methylpyrrolidin-2-ylmethyl or N-methylmorpholin-3-ylmethyl. AM-1220 was first detected as an ingredient of synthetic cannabis smoking blends in 2010.
Legal Status
As of October 2015 AM-1220 is a controlled substance in China.
