 | ||
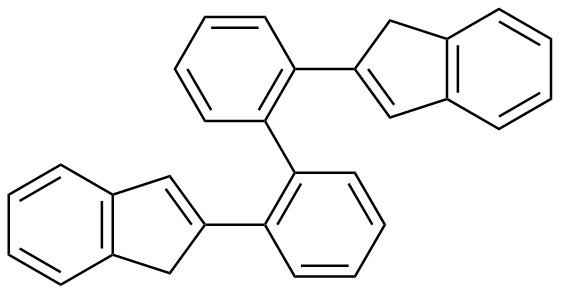
2,2'-Bis(2-indenyl) biphenyl is a ligand used to make organometallic complexes involving two aromatic-stabilized cyclopentadienyl-type carbocycle-to-metal atom bonds, complexes with distinct shapes and catalytic properties. The symmetry of the molecule--apparent in corresponding "top" and "bottom" halves of the molecule, connected together by a carbon-carbon bond between the two benzene rings (phenyl rings, see picture)--allows the two five-membered rings to participate in bonding on opposite sides of a metal atom, creating a type of sandwich compound. A prerequisite for the bonding is deprotonation of the cyclopentadiene-type five-membered ring to form the two anionic carbocycles that bind to the metal atom.
Metals studied with 2,2'-bis(2-indenyl) biphenyl include titanium, zirconium, and hafnium. The ligand and its complexes were originally prepared by the research group of the late Brice Bosnich at The University of Chicago. Zirconium and hafnium complexes made from this ligand were found to be active catalysts for the polymerization of the smallest alkenes--compounds with carbon-carbon double bonds--namely, ethylene and propylene. The use of such complexes in the polymerisation of alkenes has since been reported, and patented by DSM Research,
