Formula C4H10O2 Density 987 kg/m³ | Molar mass 90.121 g/mol | |
 | ||
Appearance nearly colorless solid or liquid | ||
2,3-Butanediol is a organic compound with the formula is (CH3)2(CHOH)2. 2,3-Butanediol has three stereoisomers, all of which are colorless, viscous liquids. Butanediols have applications as precursors to various plastics and pesticides.
Contents
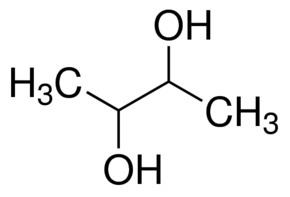
Isomerism
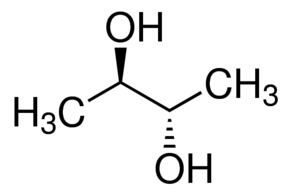
Of the three stereoisomers, two are enantiomers (levo- and dextro-2,3-butanediol) and one is a meso compound. The enantiomeric pair have (2R, 3R) and (2S, 3S) configurations at carbons 2 and 3, while the meso compound has configuration (2R, 3S) or, equivalently, (2S, 3R).
Industrial production and uses
2,3-Butanediol is prepared by hydrolysis of 2,3-butene oxide:
(CH3CH)2O + H2O → (CH3)2(CHOH)2The isomer distribution depends on the stereochemistry of the epoxide.
The meso isomer is used to combine with naphthalene-1,5-diisocyanate. The resulting polyurethane is called "Vulkollan".
Biological production
The (2R,3R)-stereoisomer of 2,3-butanediol is produced by a variety of microorganisms in a process known as butanediol fermentation. It is found naturally in cocoa butter, in the roots of Ruta graveolens, sweet corn, and in rotten mussels. It is used in the resolution of carbonyl compounds in gas chromatography.
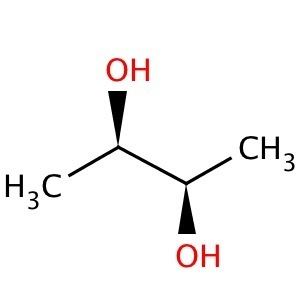
During World War II research was done towards producing 2,3-butanediol by fermentation in order to produce 1,3-butadiene, the monomer of the polybutadiene used in a leading type of synthetic rubber.
