Abbreviations TCDD; TCDBD Molar mass 321.97 g/mol Melting point 305 °C | Formula C12H4Cl4O2 Density 1.8 g/cm³ Soluble in Water | |
 | ||
Appearance colorless to white crystalline solid IUPAC ID 2,3,7,8-tetrachlorodibenzo[b,e][1,4]-dioxin | ||
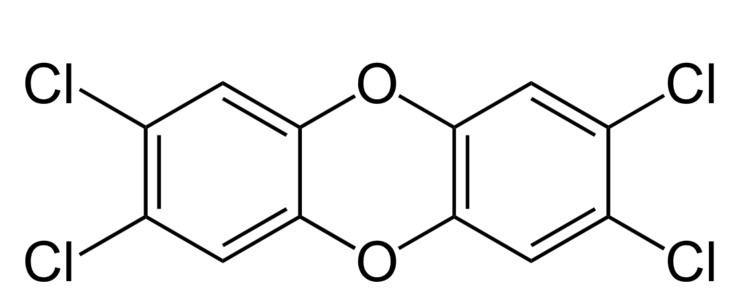
2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) is a polychlorinated dibenzo-p-dioxin (sometimes shortened, though inaccurately, to simply "dioxin") with the chemical formula C
12H
4Cl
4O
2. TCDD is a colorless solid with no distinguishable odor at room temperature. It is usually formed as a side product in organic synthesis and burning of organic materials.
Contents
- Toxicology
- Cancer
- Mechanism of action
- Teratogenic effects
- Epidemiology
- Other animals
- Developmental effects
- Cases of exposure
- References
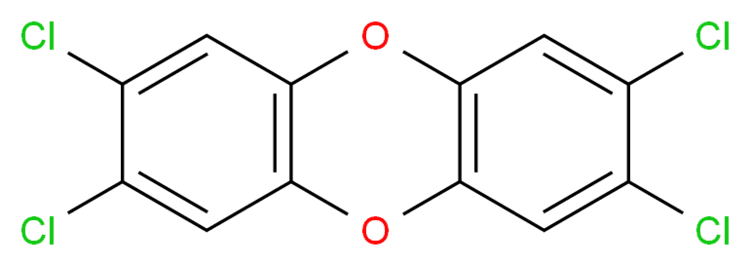
TCDD is the most potent compound (congener) of its series (polychlorinated dibenzodioxins, known as PCDDs or simply dioxins) and became known as a contaminant in Agent Orange, a herbicide used in the Vietnam War. TCDD was released into the environment in the Seveso disaster. It is a persistent environmental contaminant usually present in a complex mixture of dioxin-like compounds, and is a carcinogen.

Toxicology
The Expert Group of the World Health Organization considered developmental toxicity as the most pertinent risk of dioxins to human beings. Because people are usually exposed simultaneously to a number of dioxin-like chemicals, a more detailed account is given at dioxins and dioxin-like compounds.
Cancer
TCDD was classified in 1997 by the International Agency for Research on Cancer as a carcinogen for humans (group 1). In the occupational cohort studies available for the classification, the risk, even at very high exposures, was weak and borderline detectable. Therefore, human data were not deemed sufficient, and the classification was, in essence, based on animal experiments and mechanistic considerations. This has been criticized as a deviation from IARC classification rules. It is much debated whether TCDD is carcinogenic only at high doses which also cause toxic damage of tissues. Moreover, a recent review concludes that, after 1997, further studies do not support an association between TCDD exposure and cancer risk. New studies include the update of Vietnam veteran studies from Ranch Hand operation, which concluded that after 30 years the results do not provide evidence of disease.
There is also direct epidemiological evidence that TCDD is not carcinogenic at low doses, and in some studies cancer risk has even decreased. This is called a J-shape dose-response, low doses decrease the risk, and only higher doses increase the risk.
Mechanism of action

TCDD and dioxin-like compounds act via a specific receptor present in all cells: the aryl hydrocarbon (AH) receptor. This receptor is a transcription factor which is involved in expression of genes; in fact it has been shown that high doses of TCDD either increase or decrease the expression of several hundred genes in rats. Genes of enzymes activating the breakdown of foreign and often toxic compounds are classic examples of such genes. TCDD increases the enzymes breaking down, e.g., carcinogenic polycyclic hydrocarbons such as benzo(a)pyrene.
These polycyclic hydrocarbons also activate the AH receptor, but less than TCDD and only temporarily. Even many natural compounds present in vegetables cause some activation of the AH receptor. This phenomenon can be viewed as adaptive and beneficial, because it protects the organism from toxic and carcinogenic substances. Excessive and persistent stimulation of AH receptor, however, leads to a multitude of adverse effects.
The physiological function of the AH receptor has been the subject of continuous research. One obvious function is to increase the activity of enzymes breaking down foreign chemicals or normal chemicals of the body as needed. There may be other functions, however, related to growth of various organs or other regulatory functions. The AH receptor is phylogenetically highly conserved transcription factor with a history of at least 500 million years, and found in all vertebrates, and its ancient analogs are important regulatory proteins even in more primitive species. In fact, knock-out animals with no AH receptor are prone to illness and developmental problems. Taken together, this implies the necessity of a basal degree of AH receptor activation to achieve normal physiological function.
While the mutagenic and genotoxic effects of TCDD are sometimes disputed and sometimes confirmed it does foster the development of cancer. Its main action in causing cancer is cancer promotion; it promotes the carcinogenicity initiated by other compounds. Very high doses may, in addition, cause cancer indirectly; one of the proposed mechanisms is oxidative stress and the subsequent oxygen damage to DNA. There are other explanations such as endocrine disruption or altered signal transduction. The endocrine disrupting activities seem to be dependent on life stage, being anti-estrogenic when estrogen is present (or in high concentration) in the body, and estrogenic in the absence of estrogen.
Teratogenic effects
In Vietnam and the United States, teratogenic or birth defects were observed in children of persons that were exposed to Agent Orange or 2,4,5-T that contained TCDD as an impurity out of the production process. However the scientific data supporting a causal link between Agent Orange/dioxin exposure and birth defects is controversial and weak, in part due to poor methodology. In 2006 Anh Duc Ngo and colleagues, of the University of Texas Health Science Center in Austin, published a meta-analysis that exposed a large amount of heterogeneity/(different findings) between studies, a finding consistent with a lack of consensus on the issue. Despite this, statistical analysis of the studies they examined resulted in data that the increase in birth defects/relative risk(RR) from exposure to agent orange/dioxin "appears" to be on the order of 3 in Vietnamese funded studies but 1.29 in the rest of the world. With a casual relationship near the threshold of statistical significance in still-births, cleft palate, and neural tube defects, with spina bifida being the most statistically significant defect. The large discrepancy in RR between Vietnamese studies and those in the rest of the world have been suggested to be due to bias in the Vietnamese studies.
Epidemiology
The World Health Organization recommends a monthly limit of 70 picograms per kilogram of body weight,or 0.07 ppt
(parts per trillion) in blood.
Other animals
By far most information on toxicity of dioxin-like chemicals is based on animal studies utilizing TCDD. Almost all organs are affected by high doses of TCDD. In short-term toxicity studies in animals the typical effects are anorexia and wasting, and even after a huge dose animals die only 1 to 6 weeks after the TCDD administration. Seemingly similar species have varying sensitivities to acute effects: lethal dose for a guinea pig is about 1 µg/kg, but to a hamster it is more than 1,000 µg/kg. A similar difference can be seen even between two different rat strains. Various hyperplastic (overgrowth) or atrophic (wasting away) responses are seen in different organs, thymus atrophy is very typical in several animal species. TCDD also affects the balance of several hormones. In some species, but not in all, severe liver toxicity is seen. Taking into account the low doses of dioxins in the present human population, only two types of toxic effects have been considered to cause a relevant risk to humans: developmental effects and cancer.
Developmental effects
Developmental effects occur at very low doses in animals. They include frank teratogenicity such as cleft palate and hydronephrosis. Development of some organs may be even more sensitive: very low doses perturb the development of sexual organs in rodents, and the development of teeth in rats. The latter is important in that tooth deformities were also seen after the Seveso accident and possibly after a long breast-feeding of babies in the 1970s and 1980s when the dioxin concentrations in Europe were about ten times higher than at present.
Cancer
Cancers can be induced in animals at many sites. At sufficiently high doses TCDD has caused cancer in all animals tested. The most sensitive is liver cancer in female rats, and this has long been a basis for risk assessment. Dose-response of TCDD in causing cancer does not seem to be linear, and there is a threshold below which it seems to cause no cancer. TCDD is not mutagenic or genotoxic, in other words, it is not able to initiate cancer, and the cancer risk is based on promotion of cancer initiated by other compounds or on indirect effects such as disturbing defense mechanisms of the body e.g. by preventing apoptosis or programmed death of altered cells. Carcinogenicity is associated with tissue damage, and it is often viewed now as secondary to tissue damage.
TCDD may in some conditions potentiate the carcinogenic effects of other compounds. An example is benzo(a)pyrene that is metabolized in two steps, oxidation and conjugation. Oxidation produces epoxide carcinogens that are rapidly detoxified by conjugation, but some molecules may escape to the nucleus of the cell and bind to DNA causing a mutation, resulting in cancer initiation. When TCDD increases the activity of oxidative enzymes more than conjugation enzymes, the epoxide intermediates may increase, increasing the possibility of cancer initiation. Thus a beneficial activation of detoxifying enzymes may lead to deleterious side effects.
Cases of exposure
There have been a number of incidents where people have been exposed to high doses of TCDD or with a combination of TCDD and other dioxin-like chemicals, including:
