Formula C10H8N2 | Boiling point 273 °C | |
 | ||
Related compounds | ||
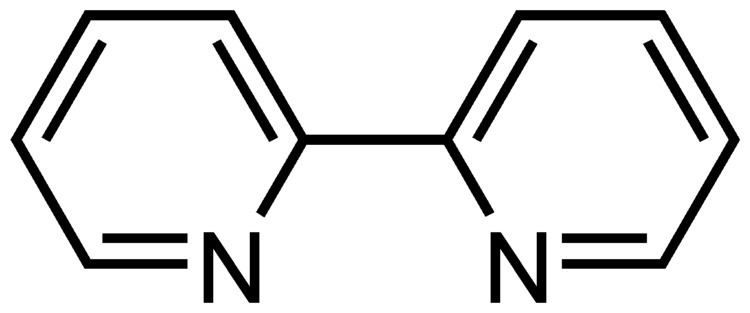
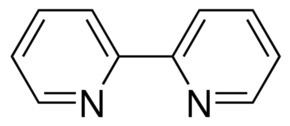
2,2′-Bipyridine (bipy or bpy, pronounced /ˈbɪpiː/) is an organic compound with the formula (C10H8N2). This colorless solid is an important isomer of the bipyridine family. It is a bidentate chelating ligand, forming complexes with many transition metals. Ruthenium complex and platinum complexes of bipy exhibit intense luminescence, which may have practical applications.
Contents
Preparation and general properties
It is prepared by the dehydrogenation of pyridine using Raney nickel:
2C5H5N → (C5H4N)2 + H2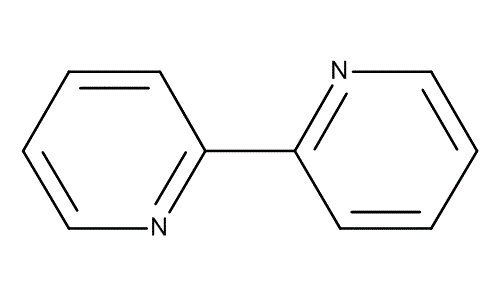
Although uncoordinated bipyridine is often drawn with its nitrogen atoms in cis conformation, the lowest energy conformation both in solid state and in solution is in fact coplanar, with nitrogen atoms in trans position. Protonated bipyridine adopts a cis conformation. Upon binding to metal ions the related N,N-heterocyclic ligand phenanthroline does not incur an enthalpic and entropic penalty, and thus its complexes tend to be more stable.
Reflecting the popularity of this ligand design, many substituted variants of bipy have been described.
Illustrative complexes

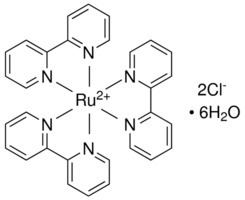
Tris-bipy complexes
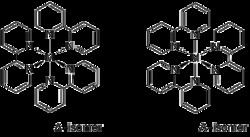
Bipyridine complexes absorb intensely in the visible part of the spectrum. The electronic transitions are attributed to metal-to-ligand charge transfer (MLCT). In the "tris(bipy) complexes" three bipyridine molecules coordinate to a metal ion, written as [M(bipy)3]n+ (M = metal ion; Cr, Fe, Co, Ru, Rh and so on; bipy = 2,2′-bipyridine). These complexes have six-coordinated, octahedral structures and two enantiomers as follows:
These and other homoleptic tris-2,2′-bipy complexes of many transition metals are electroactive. Often, both the metal centred and ligand centred electrochemical reactions are reversible one-electron reactions that can be observed by cyclic voltammetry. Under strongly reducing conditions, most tris(bipy) complexes can be reduced to neutral derivatives containing bipy− ligands. Examples include M(bipy)3, where M = Al, Cr, Si.