Formula C6H4Cl2 | ||
 | ||
Appearance Colorless/white crystals Related compounds | ||
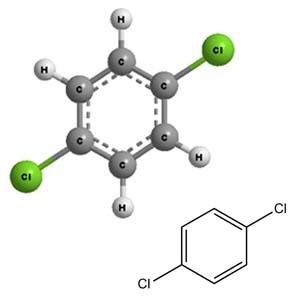
1,4-Dichlorobenzene (p-DCB or para-dichlorobenzene, sometimes abbreviated as PDB or para) is an organic compound with the formula C6H4Cl2. This colorless solid has a strong odor. The molecule consists of a benzene ring with two chlorine atoms (replacing hydrogen atoms) on opposing sites of the ring. It is used as a pesticide and a deodorant, most familiarly in mothballs in which it is a replacement for the more traditional naphthalene because of its flammability (though both chemicals have the same NFPA 704 rating). It is also used as a precursor in the production of the polymer poly(p-phenylene sulfide).
Contents
- Production
- Disinfectant deodorant and pesticide
- Precursor to other chemicals
- Environmental and health effects
- References
Production
p-DCB is produced by chlorination of benzene using ferric chloride as a catalyst:
C6H6 + 2 Cl2 → C6H4Cl2 + 2 HCl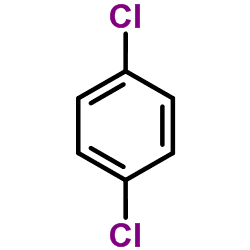
The chief impurity is the 1,2 isomer. The compound can be purified by fractional crystallisation, taking advantage of its relatively high melting point of 53.5 °C; the isomeric dichlorobenzenes and chlorobenzene melt well below room temperature.
Disinfectant, deodorant, and pesticide
p-DCB is used to control moths, molds, and mildew. It also finds use as a disinfectant in waste containers and restrooms and is the characteristic smell associated with urinal cakes. Its usefulness for these applications arises from p-DCB's low solubility in water and its relatively high volatility: it sublimes readily near room temperature.
Precursor to other chemicals
Nitration gives 1,4-dichloronitrobenzene, a precursor to commercial dyes and pigments. The chloride sites on p-DCB can be substituted with hydroxylamine and sulfide groups. In a growing application, p-DCB is the precursor to the high performance polymer poly(p-phenylene sulfide):
Environmental and health effects
p-DCB is poorly soluble in water and is not easily broken down by soil organisms. Like many hydrocarbons, p-DCB is lipophilic and will accumulate in the fatty tissues.
The United States Department of Health and Human Services (DHHS) and the International Agency for Research on Cancer (IARC) have determined that p-DCB may reasonably be anticipated to be a carcinogen. This has been indicated by animal studies, although a full-scale human study has not been done. Animals given very high levels in water developed liver and kidney tumors.
The United States Environmental Protection Agency (EPA) has set a target maximum contaminant level of 75 micrograms of p-DCB per liter of drinking water (75 μg/L), but publishes no information on the cancer risk. p-DCB is also an EPA-registered pesticide. The United States Occupational Safety and Health Administration (OSHA) has set a maximum level of 75 parts of p-DCB per million parts air in the workplace (75 ppm) for an 8-hour day, 40-hour workweek.
Under California's Proposition 65, p-DCB is listed as "known to the State to cause cancer". A probable mechanism for the carcinogenic effects of mothballs and some types of air fresheners containing p-DCB has been identified.
