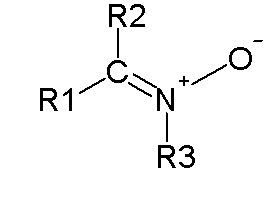
In organic chemistry, a 1,3-dipolar compound or 1,3-dipole is a dipolar compound with delocalized electrons and a separation of charge over three atoms. They are reactants in 1,3-dipolar cycloadditions.
Known 1,3-dipoles are:
Azides (RN3)Ozone (O3)Nitro compounds (RNO2)Diazo compounds (R2CN2)Some oxidesAzoxide compounds (RN(O)NR)Carbonyl oxides (Criegee zwitterions)Nitrile oxides (RCN-O)Nitrous oxide (N2O)Nitrones (R2CN(R)O)Some imines:Azomethine imineNitrilimines (RCN-NR, analogous to nitrile oxide)Carbonyl iminesSome ylidesAzomethine ylideNitrile ylide (RCNCR'2)Carbonyl ylideThiosulfines (R2CSS)